Pharmacokinetics & Pharmacodynamics of Antimicrobial Agents
2026-03-01
Pharmacokinetics and Pharmacodynamics
of Anti-infective Agents
Prof. Russell E. Lewis
Department of Molecular Medicine
University of Padua
russelledward.lewis@unipd.it
https://github.com/Russlewisbo
slides available at: www.padovaid.com
 |
Learning objectives
After completing this lecture, learners will be able to:
- Define pharmacokinetics and pharmacodynamics in the context of antiinfective therapy
- Explain the key pharmacokinetic parameters (ADME) and their clinical relevance
- Identify the three primary PK-PD indices and their associated antibiotic classes
- Compare concentration-dependent vs. time-dependent killing patterns
- Apply extended-interval and continuous infusion dosing strategies
- Discuss therapeutic drug monitoring principles for antiinfectives
- Apply PK-PD concepts to clinical case scenarios
Lecture outline
Part 1: Foundations (~30 min)
- Introduction to PK-PD
- Pharmacokinetic principles (ADME)
- Key PK parameters
- Measuring antimicrobial potency
Part 2: Applications (~45 min)
- PK-PD indices
- Killing patterns
- Dosing strategies
- TDM principles
- Clinical case studies
Why PK-PD Matters: A clinical vignette
Case Introduction
A 65-year-old man with Pseudomonas aeruginosa pneumonia is started on piperacillin-tazobactam 4.5g IV q8h.
On day 3, he’s not improving. The MIC comes back as 16 mg/L (susceptible breakpoint).
What would you do?
Options to consider:
- Increase the dose?
- Change the antibiotic?
- Change how you give the antibiotic?
We’ll return to this case at the end of the lecture…
PART 1: FOUNDATIONS
Pharmacokinetic and pharmacodynamic principles
What is pharmacology?
Definition
Pharmacology: The study concerning a compound related to its history, source, physical and chemical properties, compounding, biochemical and physiologic effects, mechanisms of action and resistance, absorption, distribution, metabolism, excretion, and therapeutic and other uses
Two key components:
- Pharmacokinetics (PK): What the body does to the drug
- Pharmacodynamics (PD): What the drug does to the body
PK vs PD: The conceptual framework
Pharmacology of antimicrobials
What makes anti-infective PK-PD unique?
Drug → Pathogen
- Direct antimicrobial effect
- Kill or inhibit growth
- Resistance selection
Drug → Host
- Efficacy
- Toxicity
- Immune modulation
Drug → Microbiome
- Collateral damage
- C. difficile risk
- Resistance reservoir
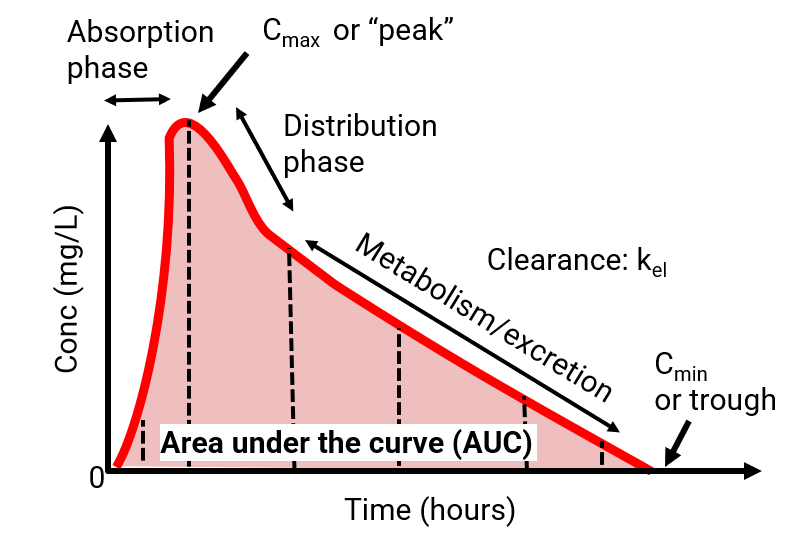
The concentration-time curve: Your roadmap
The concentration-time curve
Key parameters from the curve:
| Parameter | Symbol | Definition |
|---|---|---|
| Peak concentration | Cmax | Highest concentration achieved |
| Trough concentration | Cmin | Lowest concentration (before next dose) |
| Area under curve | AUC | Total drug exposure over time |
| Half-life | t½ | Time for concentration to decrease by 50% |
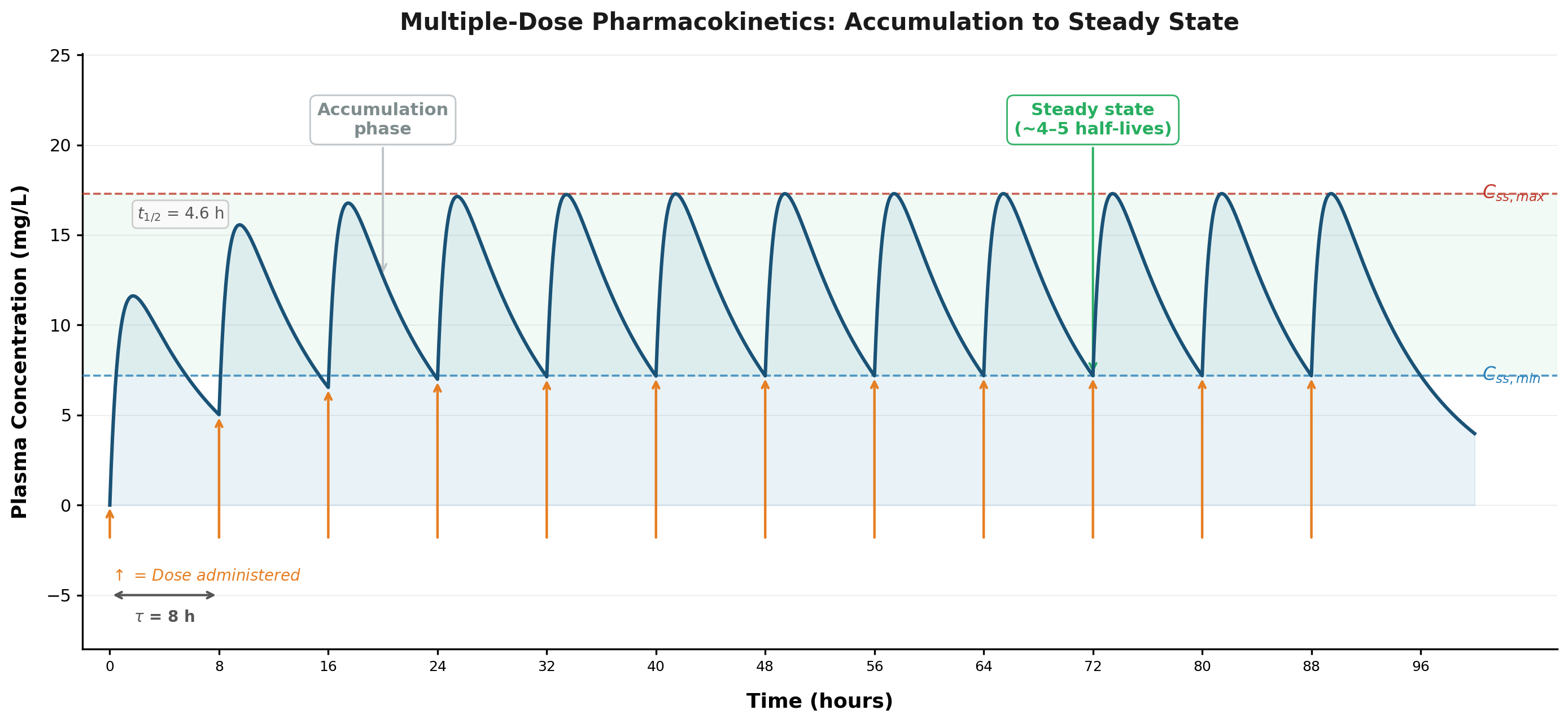
Understanding half-life (t½)
Half-life (t½) = Time required for the plasma concentration to decrease by 50%
Clinical implications:
- Time to steady state = 4-5 half-lives
- Washout time = 4-5 half-lives
- Dosing interval often based on t½
| Drug | Half-life | Typical Dosing |
|---|---|---|
| Piperacillin | 1 hour | q6-8h |
| Ceftriaxone | 8 hours | q24h |
| Azithromycin | 68 hours | Once daily × 3-5 days |
Steady state concept
Key principles:
- Accumulation occurs with repeated dosing
- Steady state = rate in equals rate out
- Takes 4-5 half-lives to achieve
- Peak and trough fluctuate around average
Clinical pearl
Don’t check “steady state” levels too early—the result won’t reflect true exposure!
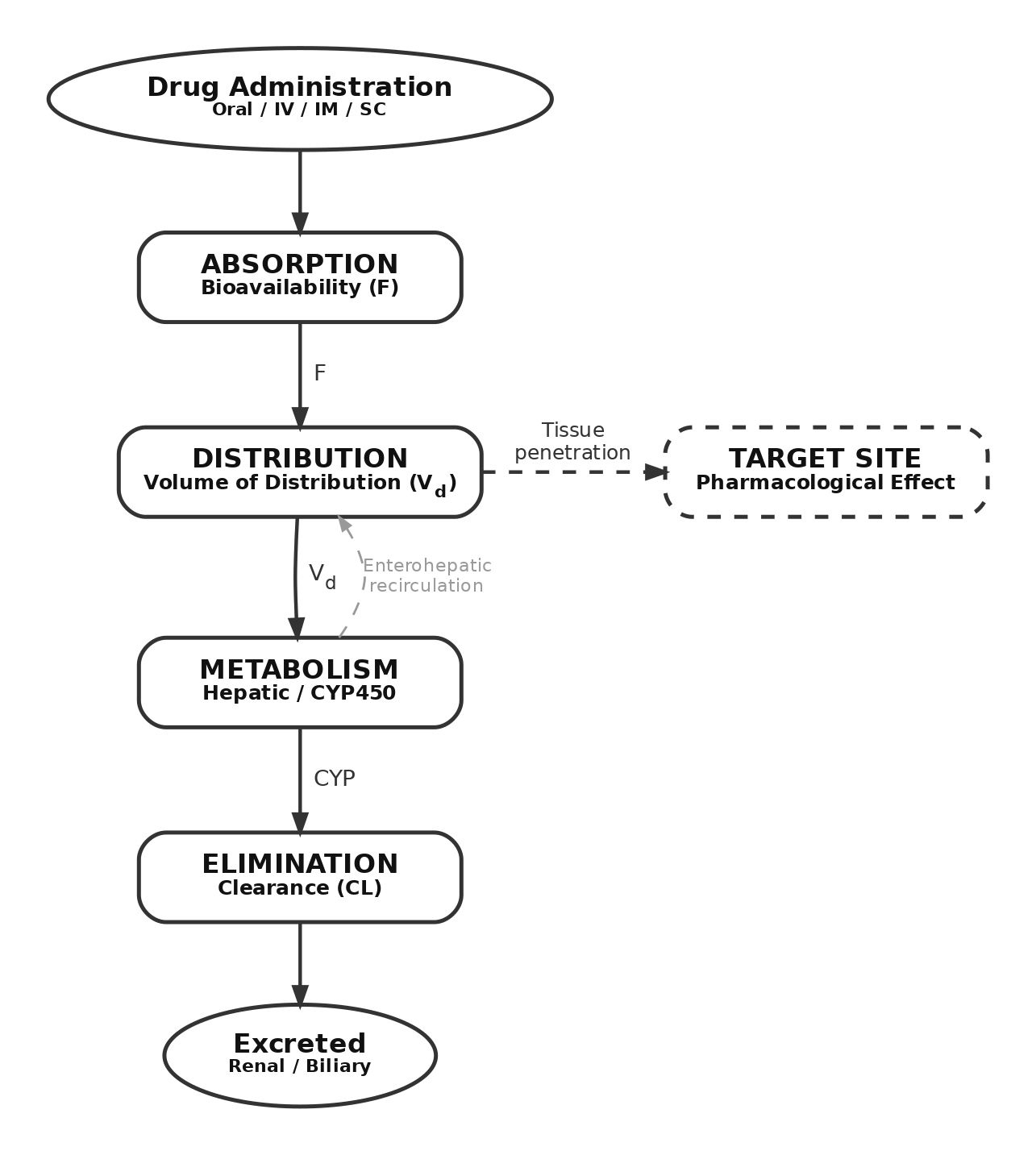
ADME: The four pillars of PK
Absorption: Getting drug into the body
Bioavailability (F)
Fraction of administered dose reaching systemic circulation
IV administration: F = 100% (by definition)
Oral administration: F varies widely
Factors affecting oral bioavailability:
- Solubility/permeability
- Gastric pH
- First-pass metabolism
- Food effects
- P-glycoprotein efflux
- Drug interactions
- Formulation
- GI disease
Clinical examples: bioavailability
| Drug | Oral Bioavailability | Clinical Note |
|---|---|---|
| Levofloxacin | ~100% | Oral = IV |
| Metronidazole | ~100% | Oral = IV |
| Amoxicillin | 70-90% | Good absorption |
| Posaconazole (suspension) | Variable | Food dependent |
| Oral vancomycin | <5% | Stays in GI tract |
IV-to-Oral conversion
High bioavailability drugs are excellent candidates for early IV-to-oral conversion—same exposure, lower cost, earlier discharge!
Enterohepatic re-circulation
Clinical significance:
- Prolongs drug elimination
- Antibiotics disrupting gut flora can interrupt this cycle
- Important for: β-lactams, mycophenolic acid, oral contraceptives
Warning
Antibiotics can reduce MPA levels in transplant patients → rejection risk!
Distribution: Where does the drug go?
Volume of distribution (Vd)
A proportionality constant relating plasma concentration to total amount of drug in the body
\[V_d = \frac{\text{Amount of drug in body}}{\text{Plasma concentration}}\]
Important concept: Vd is NOT a physiologic volume!
| Vd Value | Interpretation |
|---|---|
| ~3 L | Confined to plasma |
| ~14 L | Extracellular fluid |
| ~42 L | Total body water |
| >42 L | Extensive tissue binding |
Factors affecting distribution
Drug properties:
- Lipophilicity
- Molecular size
- Charge/ionization
- Protein binding affinity
Patient factors:
- Body composition
- Plasma proteins
- Blood flow
- Tissue barriers (BBB, prostate)
| Drug Type | Binding Protein | Effect on Vd |
|---|---|---|
| Acidic (β-lactams) | Albumin | Lower Vd |
| Basic (macrolides) | α₁-acid glycoprotein | Higher Vd |
Protein binding: Clinical significance
Only UNBOUND drug is pharmacologically active!
Why it matters:
- MIC testing uses unbound drug
- Highly bound = less active drug
- Free drug crosses membranes
- Free drug is eliminated
Clinical scenarios:
- Hypoalbuminemia → ↑ free drug
- Uremia → ↓ protein binding
- Drug displacement interactions
- Burns, sepsis, cirrhosis
Example: Ceftriaxone
85-95% protein bound → only 5-15% is active. In hypoalbuminemia, free fraction increases → potential toxicity and altered PK
Metabolism: Phase I reactions
Cytochrome P450 system
Heme-containing enzymes in the liver (and gut) that oxidize drugs
Primary CYP enzymes (by importance for drugs):
- CYP3A4 ← Metabolizes ~50% of drugs
- CYP2D6
- CYP2C9/2C19
- CYP1A2
- CYP2E1
CYP nomenclature: CYP3A4 = Family 3, Subfamily A, Gene 4
Genetic polymorphism in drug metabolism
CYP2C19 Example
Polymorphisms create distinct metabolizer phenotypes:
| Phenotype | Frequency | Clinical Effect |
|---|---|---|
| Poor metabolizer | 2-5% Caucasians, 15-20% Asians | ↑ Drug levels |
| Intermediate metabolizer | 25-35% | Moderately ↑ levels |
| Extensive metabolizer | 35-50% | Normal metabolism |
| Ultrarapid metabolizer | 5-10% | ↓ Drug levels |
Voriconazole: CYP2C19 poor metabolizers have 4× higher exposure → toxicity risk
Clinical application: PK boosting
Examples of boosted regimens:
- Lopinavir/ritonavir (Kaletra)
- Darunavir/ritonavir or /cobicistat
- Nirmatrelvir/ritonavir (Paxlovid)
Elimination: Renal clearance
Total Clearance = Renal + Nonrenal
Renal elimination mechanisms:
- Glomerular filtration
- Passive, depends on GFR
- Only unbound drug filtered
- Tubular secretion
- Active transport (OAT, OCT)
- Can exceed GFR
- Tubular reabsorption
- Returns drug to circulation
Primarily renally cleared:
- Aminoglycosides
- Vancomycin
- β-lactams (most)
- Fluoroquinolones (partial)
- Acyclovir
- Fluconazole
Elimination: Nonrenal Clearance
Nonrenal routes:
- Hepatic metabolism (most common)
- Biliary excretion (ceftriaxone, azithromycin)
- Intestinal secretion
- Other (lung, skin)
| Drug | Primary Elimination | Dose Adjustment |
|---|---|---|
| Ceftriaxone | 40% biliary | None for renal impairment |
| Metronidazole | Hepatic | Reduce in liver failure |
| Azithromycin | Biliary/fecal | None for renal impairment |
Measuring antimicrobial potency: The MIC
Minimum Inhibitory Concentration (MIC)
The lowest concentration of an antimicrobial that inhibits visible growth of a microorganism after overnight incubation
Key points:
- Measured using serial 2-fold dilutions (log₂ scale)
- Values: 0.25, 0.5, 1, 2, 4, 8, 16… mg/L
- MIC90: MIC for 90% of isolates tested
- Does NOT measure killing—only inhibition
Limitations of In vitro parameters
What an MIC doesn’t tell us…
- Rate of killing at different concentrations
- Persistent effects after drug removal (PAE)
- Immune system interactions
- Resistance emergence over time
- Tissue penetration and local concentrations
- Inoculum effects (higher bacterial loads)
The Solution: PK-PD Integration
Combine MIC with pharmacokinetic parameters to predict clinical outcomes
PK/PD parameters
PART 2: APPLICATIONS
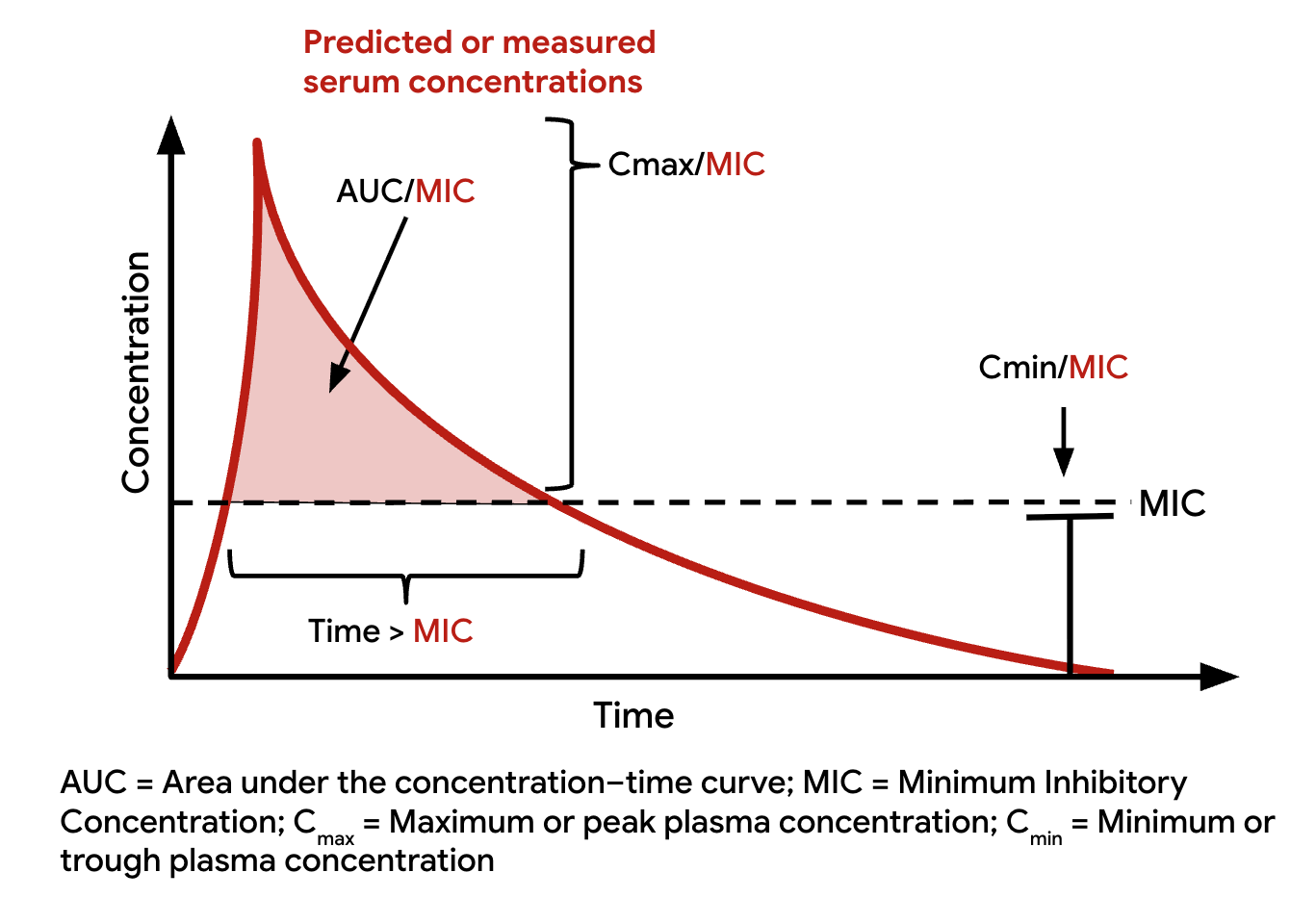
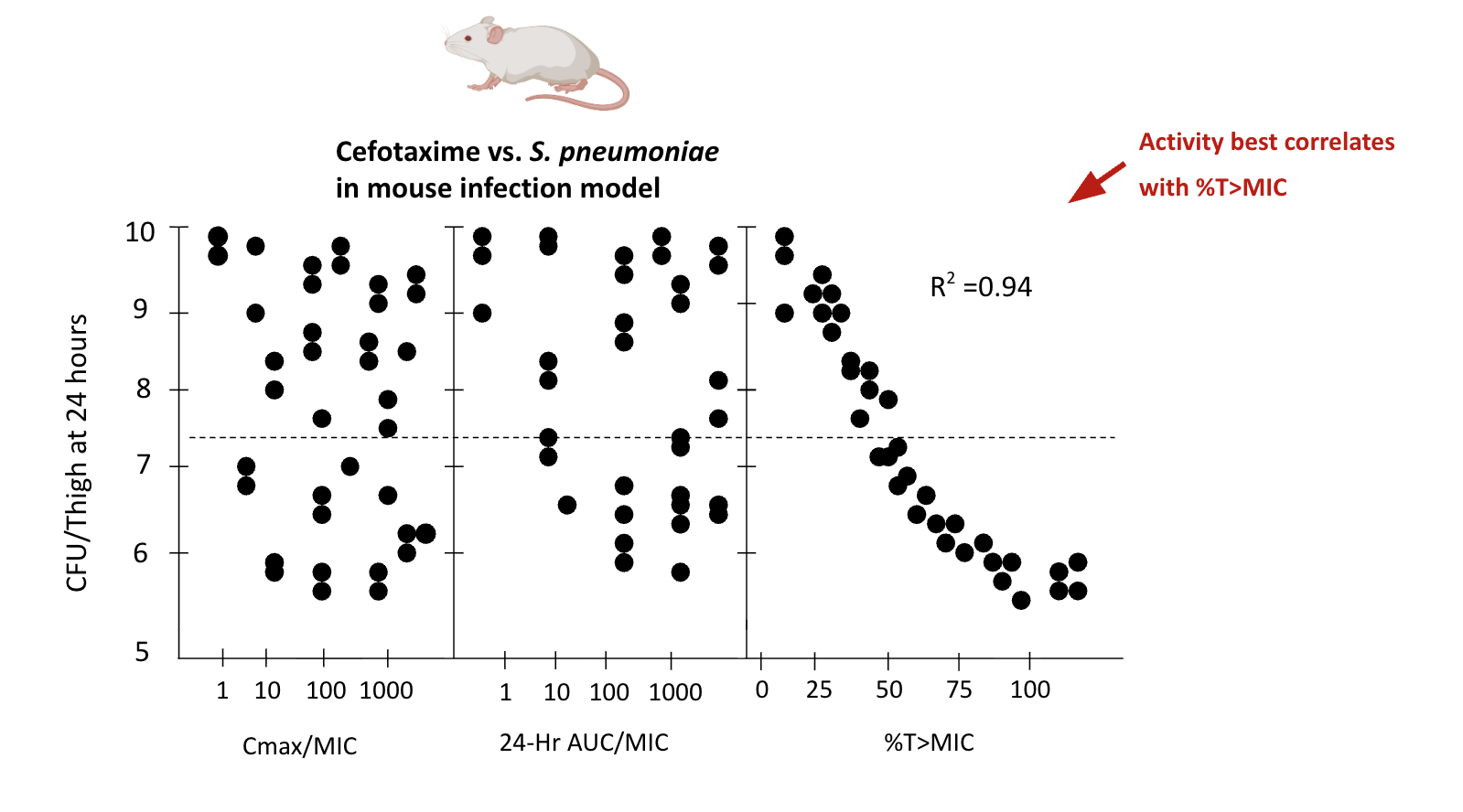
The three PK-PD indices
| Index | Formula | What It Measures |
|---|---|---|
| Cmax/MIC | Peak / MIC | Intensity of exposure |
| AUC/MIC | AUC₀₋₂₄ / MIC | Total exposure relative to potency |
| T > MIC | % time above MIC | Duration of effective exposure |
Key Insight
The “best” index depends on the antibiotic’s mechanism of action and killing characteristics. May be reported as total drug or fraction unbound (FU)- i.e. non protein bound drug
Example: Cefotaxime in animal models
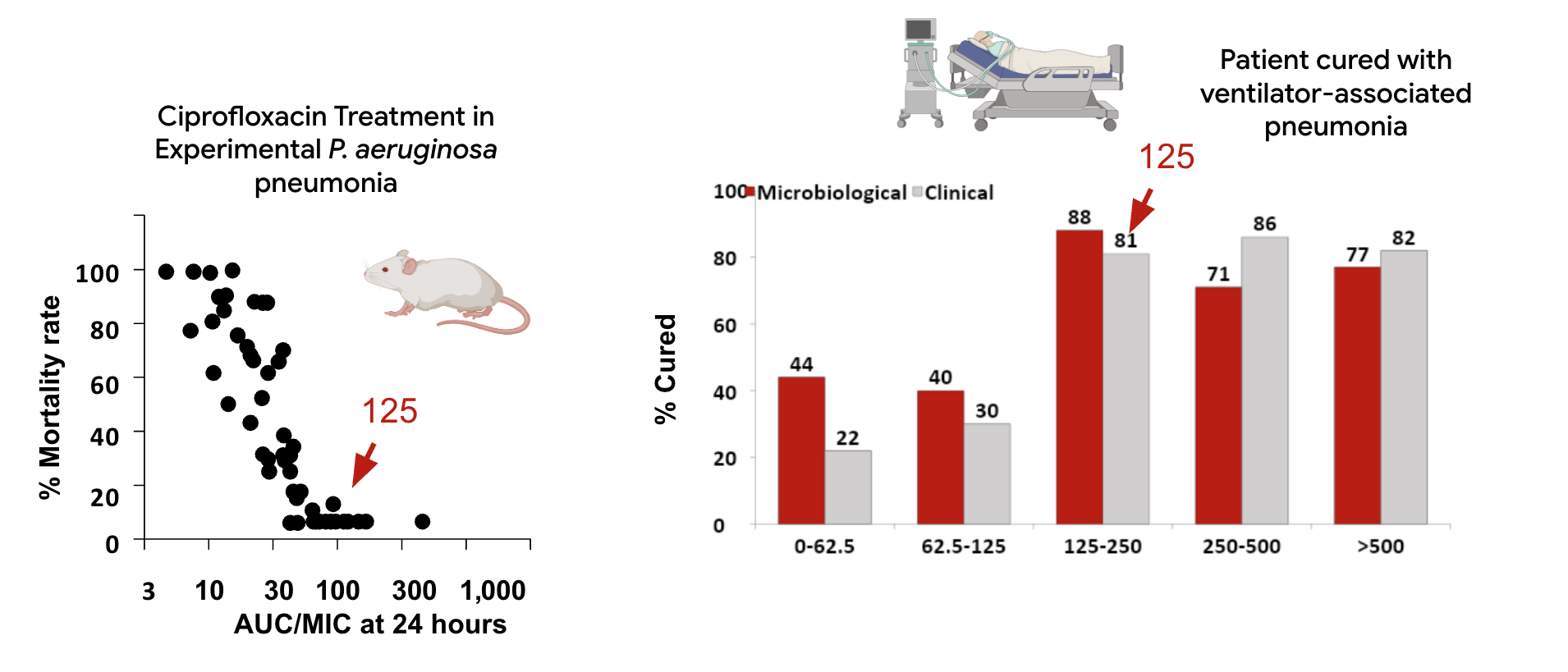
Example: Ciprofloxacin in mice vs. humans
PK-PD index by antibiotic class
| Antibiotic Class | Primary Index | Target |
|---|---|---|
| Aminoglycosides | Cmax/MIC | 8-10 |
| Fluoroquinolones | AUC/MIC (or Cmax/MIC) | 30-50 (Gram+), 100-125 (Gram-) |
| β-Lactams | T > MIC | 40-70% of interval |
| Vancomycin | AUC/MIC | 400-600 |
| Daptomycin | AUC/MIC (or Cmax/MIC) | Variable |
| Linezolid | AUC/MIC | 80-120 |
Concentration-dependent killing
Characteristics:
- Killing rate increases with concentration
- Significant post-antibiotic effect (PAE)
- Optimal strategy: Maximize the peak
Agents:
- Aminoglycosides
- Fluoroquinolones
- Daptomycin
- Metronidazole
- Amphotericin B
Concentration-dependent killing: The evidence
Classic Study: Blaser et al.
Examined Cmax/MIC ratios for enoxacin and netilmicin against Gram-negative organisms:
| Cmax/MIC Ratio | Outcome |
|---|---|
| < 8 | Bacterial regrowth in ALL cultures |
| ≥ 8 | Sustained killing |
Critical finding: When antibiotics were redosed after regrowth at sub-optimal ratios, NO killing occurred due to resistance emergence
Time-dependent killing
Duration above MIC = Efficacy
Characteristics:
- Killing rate saturates at low multiples of MIC (4×)
- Minimal post-antibiotic effect (against Gram-negatives)
- Optimal strategy: Maximize time above MIC
Agents:
- Penicillins
- Cephalosporins
- Carbapenems
- Aztreonam
- Macrolides, clindamycin
- Linezolid
Time-dependent killing: Animal model evidence
Classic Data: S.pneumoniae studies
Treatment with penicillins or cephalosporins showed dramatic mortality differences:
| T > MIC | Mortality |
|---|---|
| ≤ 20% of dosing interval | 100% |
| 40-50% of dosing interval | 0-10% |
Target T > MIC:
- Bacteriostatic effect: 30-40%
- Bactericidal effect: 50-70%
- Maximum effect: ≥70%
Post-Antibiotic Effect (PAE)
Definition
Suppression of bacterial growth that persists after drug concentrations fall below MIC
| Antibiotic Class | PAE: Gram-negative | PAE: Gram-positive |
|---|---|---|
| Aminoglycosides | 2-6 hours | 2 hours |
| Fluoroquinolones | 2-6 hours | 2 hours |
| Carbapenems | 1-2 hours | 2 hours |
| Penicillins | Little/none | 2 hours |
| Cephalosporins | Little/none | 2 hours |
PAE: Clinical implications
Long PAE:
- Allows extended dosing intervals
- Supports once-daily dosing
- Example: Once-daily aminoglycosides
Short/No PAE:
- Needs sustained concentrations
- Consider continuous infusion
- Example: β-lactams via extended infusion
PAE enhancement
PAE can be prolonged by: - Higher drug concentrations - Longer exposure duration - Sub-inhibitory concentrations
Methods for studying PK-PD
Three complementary approaches:
- In vitro models (Hollow fiber)
- Animal models (Neutropenic mouse)
- Clinical studies (Human PK-PD)
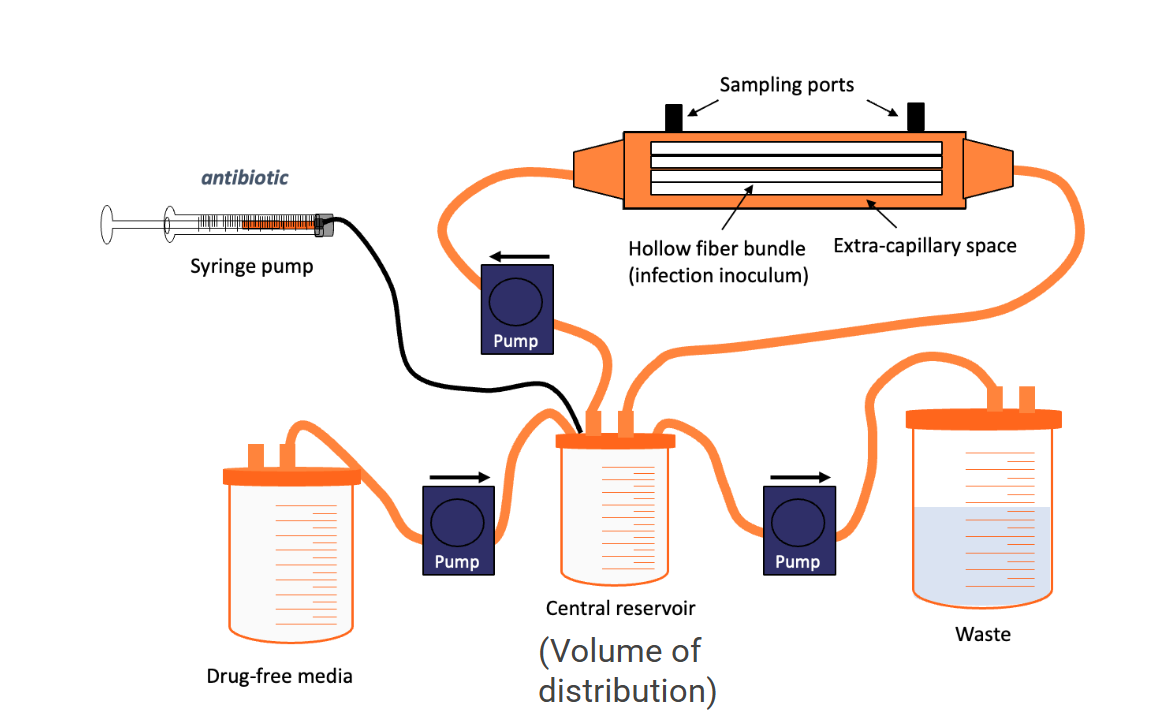
Hollow fiber infection model
Hollow fiber infection model
Hollow fiber infection model
How it works:
- Bacteria grow in extracapillary space
- Drug pumped through fibers simulates human PK
- Serial sampling measures bacterial counts
- Can model resistance emergence
Advantages:
- Precise control of drug concentrations
- Mimics human PK profiles
- No animal subjects
Limitations:
- No immune system
- May overestimate required doses
Translating PK-PD to clinical dosing
From bench to bedside:
- Identify the PK-PD index that predicts efficacy
- Determine the target value from preclinical studies
- Use Monte Carlo simulation to find doses achieving target in most patients
- Validate in clinical trials
- Apply to patient populations
Challenge
Population average doses may not achieve targets in all patients—especially critically ill!
Extended-interval aminoglycoside dosing
The revolution in aminoglycoside dosing!
Traditional: 1-2 mg/kg q8h → Modern: 5-7 mg/kg q24h
| Parameter | Traditional | Extended-Interval |
|---|---|---|
| Peak (Cmax) | 5-10 mg/L | 15-25 mg/L |
| Trough | <2 mg/L | <1 mg/L |
| Cmax/MIC | Often <8 | 8-10 |
| Nephrotoxicity | Higher | Lower |
| Efficacy | Variable | Optimized |
Extended-interval aminoglycosides: Rationale
Why it works:
- Concentration-dependent killing: Higher peaks = better killing
- Post-antibiotic effect: Covers drug-free interval
- Adaptive resistance: Drug-free time allows resensitization
- Saturable renal uptake: Less accumulation in renal cortex
Target
Achieve Cmax/MIC of 8-10 based on expected MIC90 of target organisms
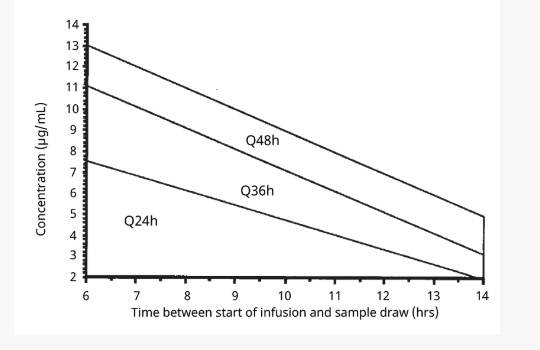
Extended interval aminoglycoside TDM
β-Lactam Infusion Strategies
| Strategy | Infusion Time | T > MIC | Daily Dose |
|---|---|---|---|
| Intermittent | 30 min | Lowest | Standard |
| Extended | 3-4 hours | Higher | Same or lower |
| Continuous | 24 hours | 100% | Often lower |
β-Lactam Infusion Strategies
Extended infusion: The loading dose
Always give a loading dose:
- Achieves therapeutic levels immediately
- Critical when bacterial load is highest
- Load = Standard intermittent dose
- Then transition to extended/continuous infusion
Clinical evidence: Extended β-Lactam infusions
| Evidence | Findings |
|---|---|
| Meta-analysis (Rhodes 2018) | 1.46× lower mortality with prolonged pip-tazo |
| Meta-analysis (Falagas 2013) | Lower mortality with prolonged carbapenems |
| BLING-II RCT | Comparable 90-day survival |
| BLISS RCT | Comparable outcomes |
Why RCTs show less benefit
- Selection bias in observational studies (sicker patients get standard dosing)
- RCTs include patients who would do well either way
- Greatest benefit likely in: high MICs, critically ill, immunocompromised
Special populations: Critically ill patients
Why PK is altered in critical illness:
↑ Volume of distribution:
- Third spacing
- Fluid resuscitation
- Hypoalbuminemia
- Capillary leak
Altered Clearance:
- Augmented renal clearance (ARC)
- Acute kidney injury
- CRRT
- Hepatic dysfunction
Important
Standard doses often underdose critically ill patients initially, then overdose as organ function changes. DALI study: More than one-third of critically Ill patients to not acheive minimum dosing targets!
Therapeutic Drug Monitoring:
Key Principles
When is TDM valuable?
- Narrow therapeutic index
- High interpatient variability
- Poorly predictable PK
- Defined PK-PD targets
- Concentration-related toxicity
- Clinical consequences of under/overdosing
Commonly monitored antiinfectives
Aminoglycosides, vancomycin, voriconazole, posaconazole, flucytosine
Vancomycin: Evolution of monitoring
Old Paradigm:
- Target: Trough 15-20 mg/L
- Simple to implement
- “Higher trough = better”
- Associated with nephrotoxicity
New Paradigm (2020 Guidelines):
- Target: AUC/MIC 400-600
- AUC-based dosing
- Requires 2 samples or Bayesian
- Better precision, less toxicity
Key Change
Troughs of 15-20 mg/L often give AUC/MIC >600 → increased nephrotoxicity without added benefit
Vancomycin AUC estimation methods
Option 1: Two-Sample Method
- Draw peak (1-2h post-infusion) and trough (before next dose)
- Calculate AUC using first-order equations
- More accurate, more complex
Option 2: Bayesian Estimation
- Single trough concentration
- Population PK model + patient covariates
- Software estimates individual PK parameters
- Predicts AUC from one level
Practical Tip
Many institutions are implementing Bayesian vancomycin dosing software (e.g., DoseMeRx, InsightRx)
Antifungal TDM: triazoles
Why TDM is important for triazoles:
| Drug | Issue | Target Range |
|---|---|---|
| Voriconazole | CYP2C19 polymorphism | 1-5 mg/L |
| Itraconazole | Variable absorption | ≥0.5-1 mg/L |
| Posaconazole | Food-dependent absorption | ≥0.7-1 mg/L |
Voriconazole toxicity
Levels >5.5 mg/L associated with:
Visual disturbances
Hepatotoxicity
CNS effects
Hallucinations
Antiretroviral PK-PD: Unique considerations
What makes ARVs different:
- Site of action: Inside mammalian cells
- Many require intracellular activation (NRTIs → triphosphates)
- Plasma levels may not reflect site concentrations
- Combination therapy is standard
The Therapeutic Window
Antiretrovirals must achieve concentrations that:
Suppress viral replication (efficacy)
Don’t cause toxicity
Prevent resistance emergence
Case Study 1: Aminoglycoside dosing
Clinical Scenario
62-year-old man, 80 kg, CrCl 90 mL/min, with hospital-acquired pneumonia. You want to start tobramycin. MIC90 of P. aeruginosa at your hospital is 2 mg/L.
Traditional dosing: 1.5 mg/kg q8h = 120 mg q8h Peak expected: ~5-6 mg/L → Cmax/MIC = 2.5-3 ❌
Extended-interval: 7 mg/kg q24h = 560 mg q24h Peak expected: ~20 mg/L → Cmax/MIC = 10 ✓
Answer: Extended-interval dosing achieves the PK-PD target of 8-10
Case study 2: β-Lactam optimization
Clinical Scenario
55-year-old woman with P. aeruginosa bloodstream infection. Piperacillin MIC = 16 mg/L (susceptible). Started on pip-tazo 4.5g q8h (30-min infusion).
Day 3: Still febrile, blood cultures remain positive.
Analysis:
- β-lactam → Time-dependent killing → T > MIC matters
- With MIC of 16 mg/L, standard dosing achieves T > MIC of only ~30%
- Target: T > MIC ≥50%
Solution: Extended infusion 4.5g q8h over 4 hours (with loading dose) OR increase to q6h dosing
Case Study 3: Vancomycin monitoring
Clinical Scenario
45-year-old man with MRSA bacteremia (MIC = 1 mg/L). Started on vancomycin 1.5g q12h. Day 3 trough = 22 mg/L.
Old approach: Trough in target range (15-20)—maybe even too high. Continue same dose?
New approach: Using Bayesian software, estimated AUC = 680 mg·h/L - AUC/MIC = 680 (target: 400-600) - This patient is overexposed → nephrotoxicity risk
Action: Reduce dose to target AUC/MIC of 400-600
Return to Opening Case
Remember Our Patient?
65-year-old man with P. aeruginosa pneumonia on pip-tazo 4.5g q8h, not improving. MIC = 16 mg/L.
Now you can answer:
- What’s the relevant PK-PD index? T > MIC (β-lactam)
- Is the target being achieved? Likely not with high MIC
- What’s your recommendation?
- Extended infusion (4.5g over 4h q8h) with loading dose
- OR increase frequency to q6h
- Consider combination therapy
- Why not just increase the dose? Extending infusion increases T > MIC more efficiently than higher doses
Summary: Key Clinical pearls
Know your PK-PD index:
Cmax/MIC: aminoglycosides, fluoroquinolones
AUC/MIC: vancomycin, fluoroquinolones
T > MIC: β-lactams 2.
Match dosing strategy to PK-PD:
Concentration-dependent → maximize peak
Time-dependent → maximize duration
Remember special populations:
Critically ill patients often need higher/more frequent doses initially
TDM helps individualize therapy
Summary: Dosing Strategy quick reference
| Drug Class | Strategy | Rationale |
|---|---|---|
| Aminoglycosides | Once-daily high dose | Optimize Cmax/MIC, minimize toxicity |
| Fluoroquinolones | Higher doses when possible | Cmax/MIC and AUC/MIC |
| β-Lactams | Extended/continuous infusion | Maximize T > MIC |
| Vancomycin | AUC-based dosing | Target AUC/MIC 400-600 |
| Azoles | TDM-guided | High variability |
Take-Home Messages
PK-PD integration is essential for optimizing anti-infective therapy
Underdosing is common and promotes resistance
One size doesn’t fit all—individualize based on patient and pathogen
Extended/continuous infusions can rescue patients failing standard β-lactam dosing
TDM is your tool for precision antiinfective therapy
Apply these principles every time you write an antibiotic order!
References
Supplementary: PK Abbreviations Reference
| Abbreviation | Definition |
|---|---|
| F | Bioavailability |
| Vd | Volume of distribution |
| CL | Clearance |
| t½ | Half-life |
| Cmax | Maximum concentration |
| Cmin | Minimum/trough concentration |
| AUC | Area under the concentration-time curve |
| MIC | Minimum inhibitory concentration |
| PAE | Post-antibiotic effect |
| T > MIC | Time above MIC |
| TDM | Therapeutic drug monitoring |