Penicillin and Antibiotic Allergies
Prof. Russell E. Lewis
Department of Molecular Medicine
University of Padua
 |
russelledward.lewis@unipd.it
https://github.com/Russlewisbo
Slides and course materials: www.padovaid.com
Objectives
Describe the different types of hypersensitivity reactions based on clinical presentation and immunological mechanisms
Recognise a patient history that will differentiate between immediate and delayed-type hypersensitivity reactions
Describe the risk of cross-reactions between various beta-lactam antibiotics
Describe the principles and contraindications for desensitisation
Describe the clinical manifestations, diagnosis and management of common non-beta-lactam antibiotic allergies
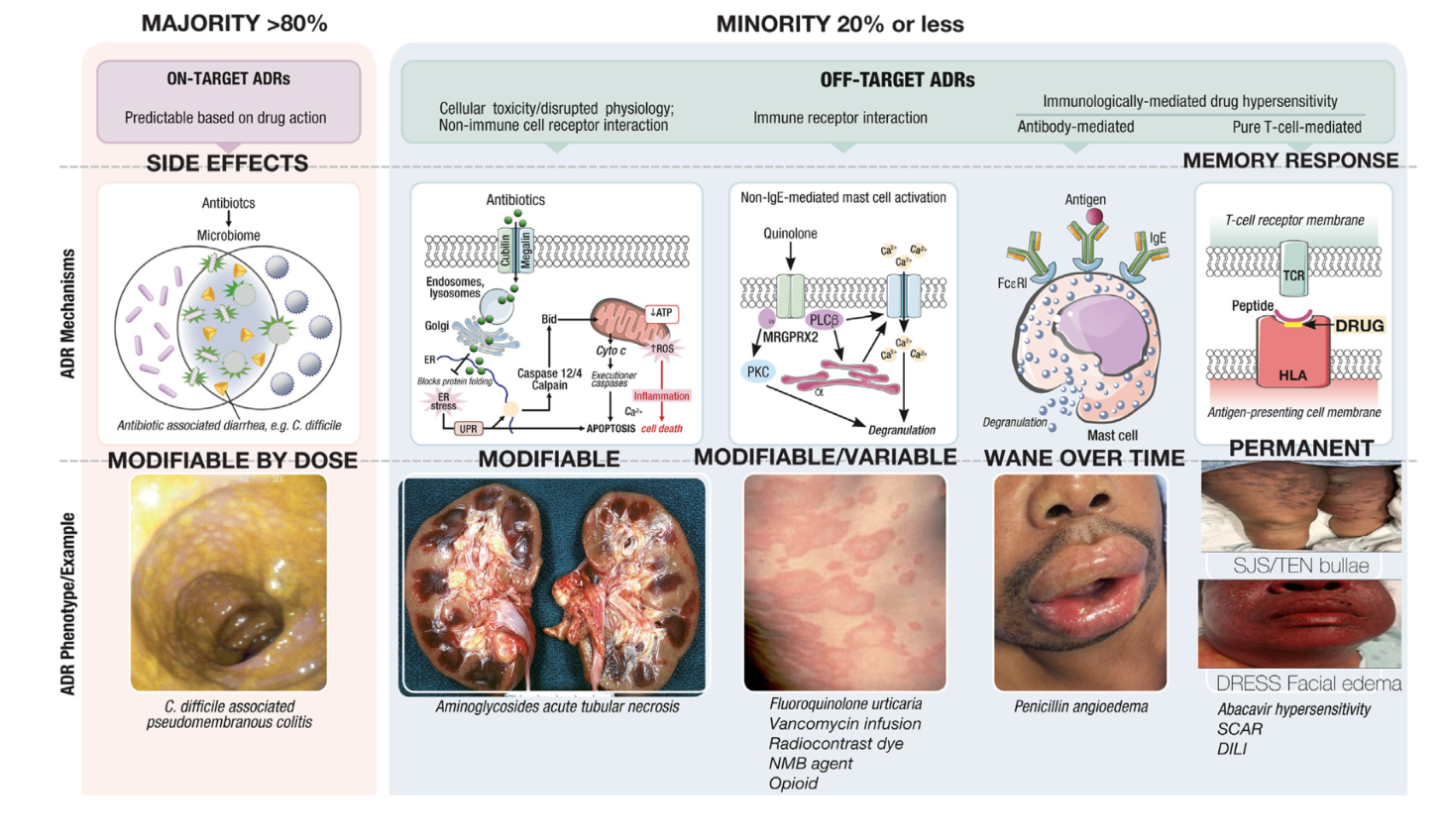
On target vs. off-target drug effects
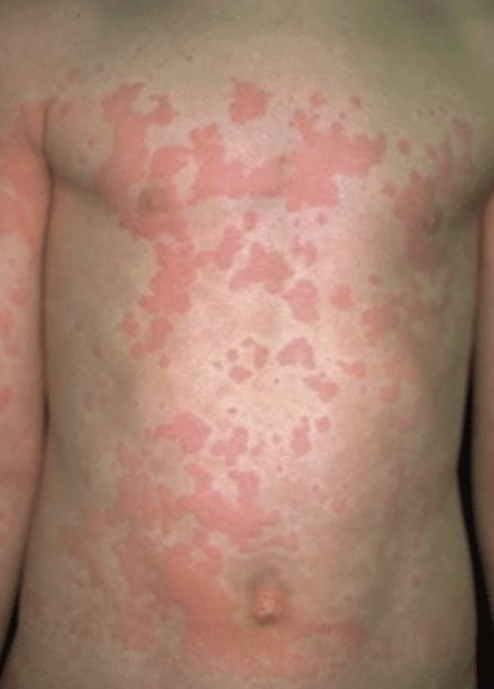
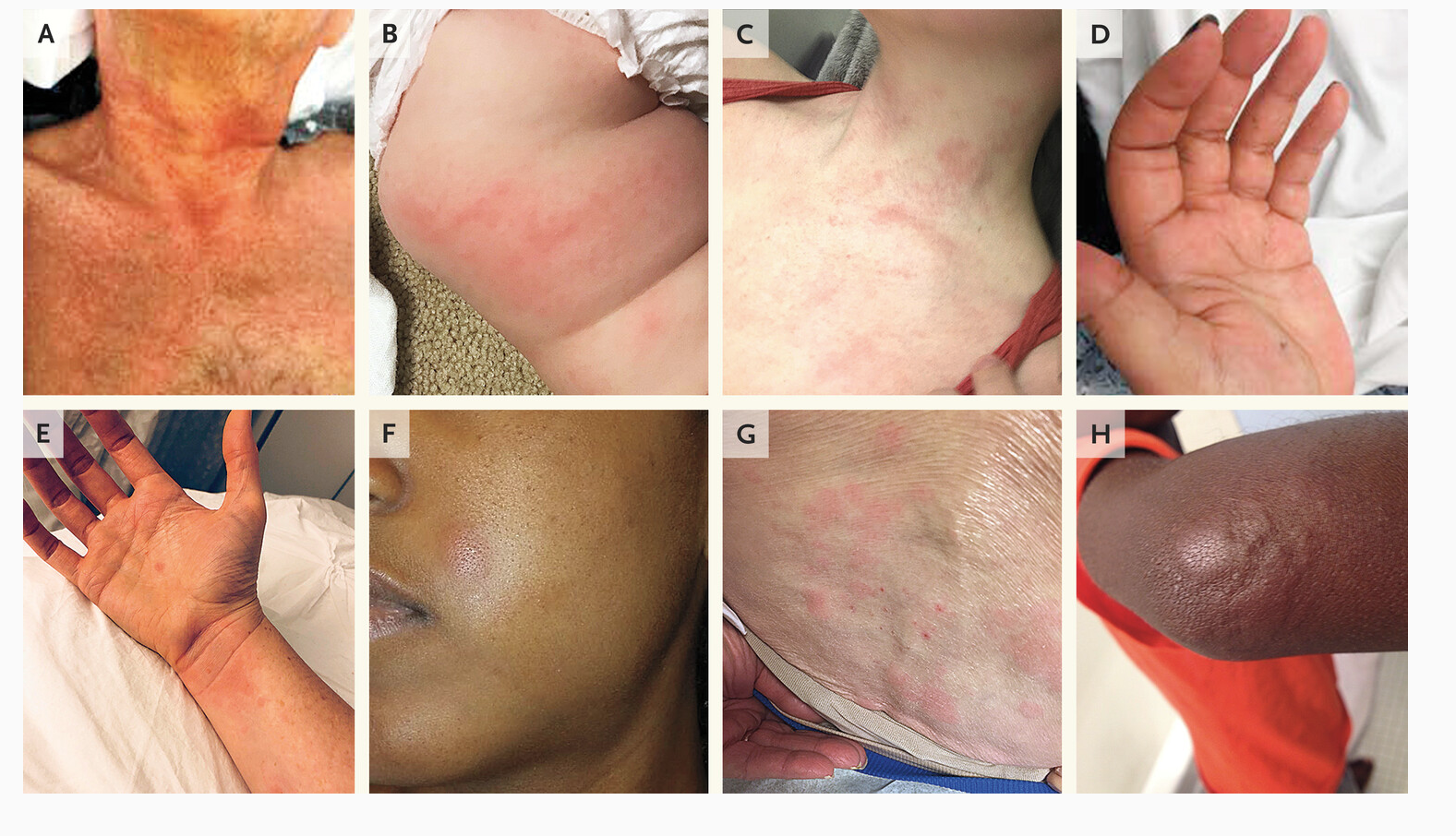
Variable cutaneous presentation of
allergic drug reactions
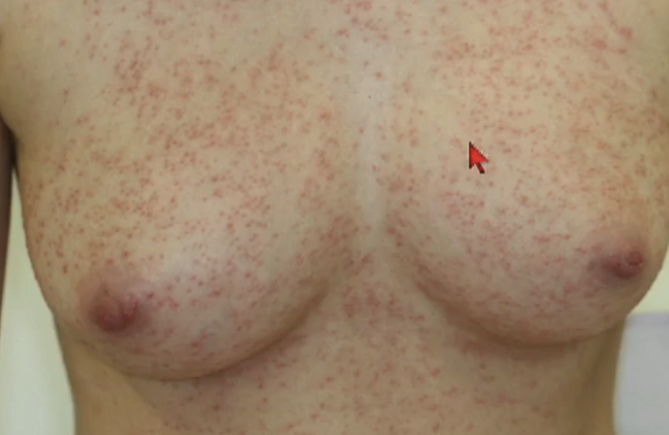
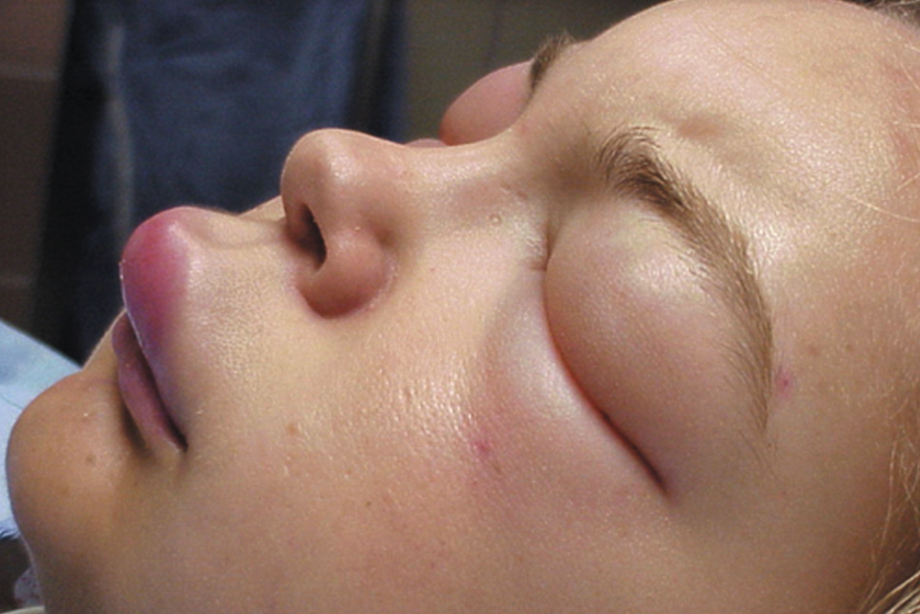
Urticaria and angioedema
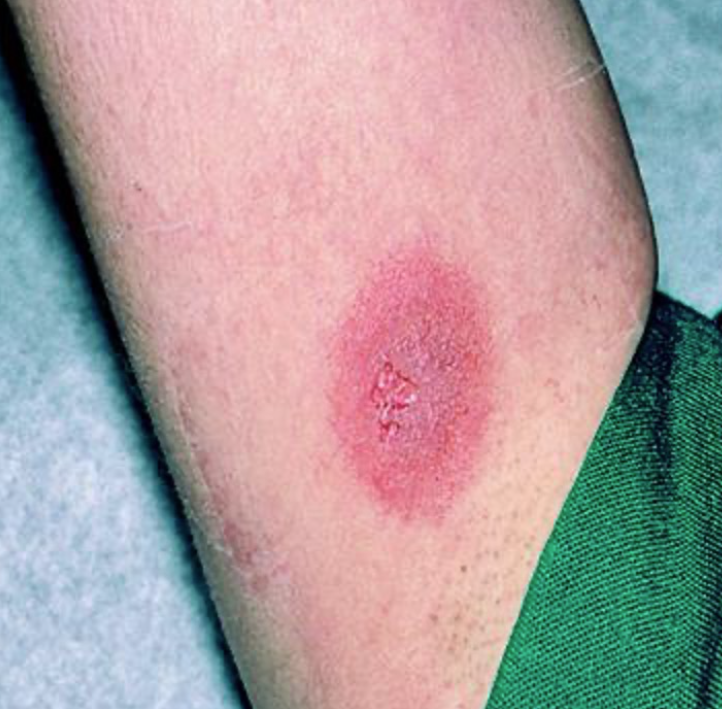
Fixed drug eruption
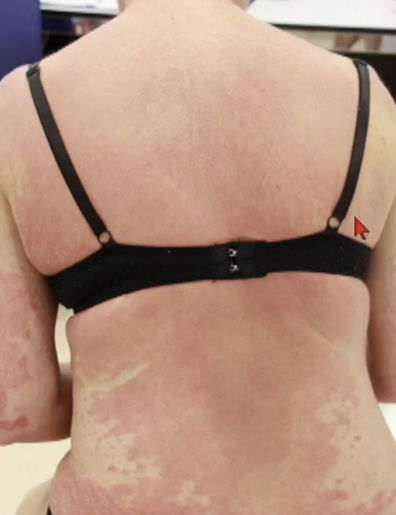
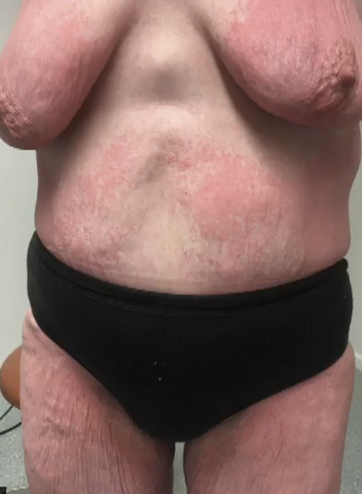
SCAR: Severe Cutaneous Adverse Reactions
Erythema multiforme,
Toxic epidermal necrolysis (TEN)
DRESS: Drug rash with eosinophilia
and systemic symptoms
Latency: 2-8 weeks
Non-specific symptoms:
- Fever (75%), lymphadenopathy (55-65%)
Hematological abnormalities:
Eosinophilia > 700 mcL (85-95%)
Leukocytosis (95%), neutrophilia (78%), monocytosis (69%)
Atypical lymphocytosis (35-67%)
Visceral involvement:
Liver (53-90%)-cholestatic and/or hepatocellular
Pulmonary (30%)-shortness of breath, cough
Cardiac involvement (2-20%)- hypotension, tachycardia, dyspnea, LV dysfunction, myocarditis
DRESS: Pathophysiology
and systemic symptoms
Pathophysiology:
Type IV T-cell activation (CD4+/CD8+) producing TNF-α
Reactivation of viruses from the Herpesviridae family (eg, HHV-6, HHV-7, Epstein-Barr virus [EBV], cytomegalovirus [CMV]) occurs in up to 75 percent of patients-cause or consequence?
Some patient human leukocyte antigens are associated with higher risk
RegiSCAR scoring system is a commonly used tool for diagnosis
Symptoms may worsen or recur despite drug discontinuation or persisnt, requing immunosuppressive treatment
Classic “high-risk” drugs for DRESS
Allopurinol
Aromatic antiepileptic agents (carbamazepine, phenytoin, lamotrigine, …)
Sulphonamides
Vancomycin
Minocycline
Nevirapine
Anti-tuberculosis drugs
Mexiletine
β-lactams are lower risk
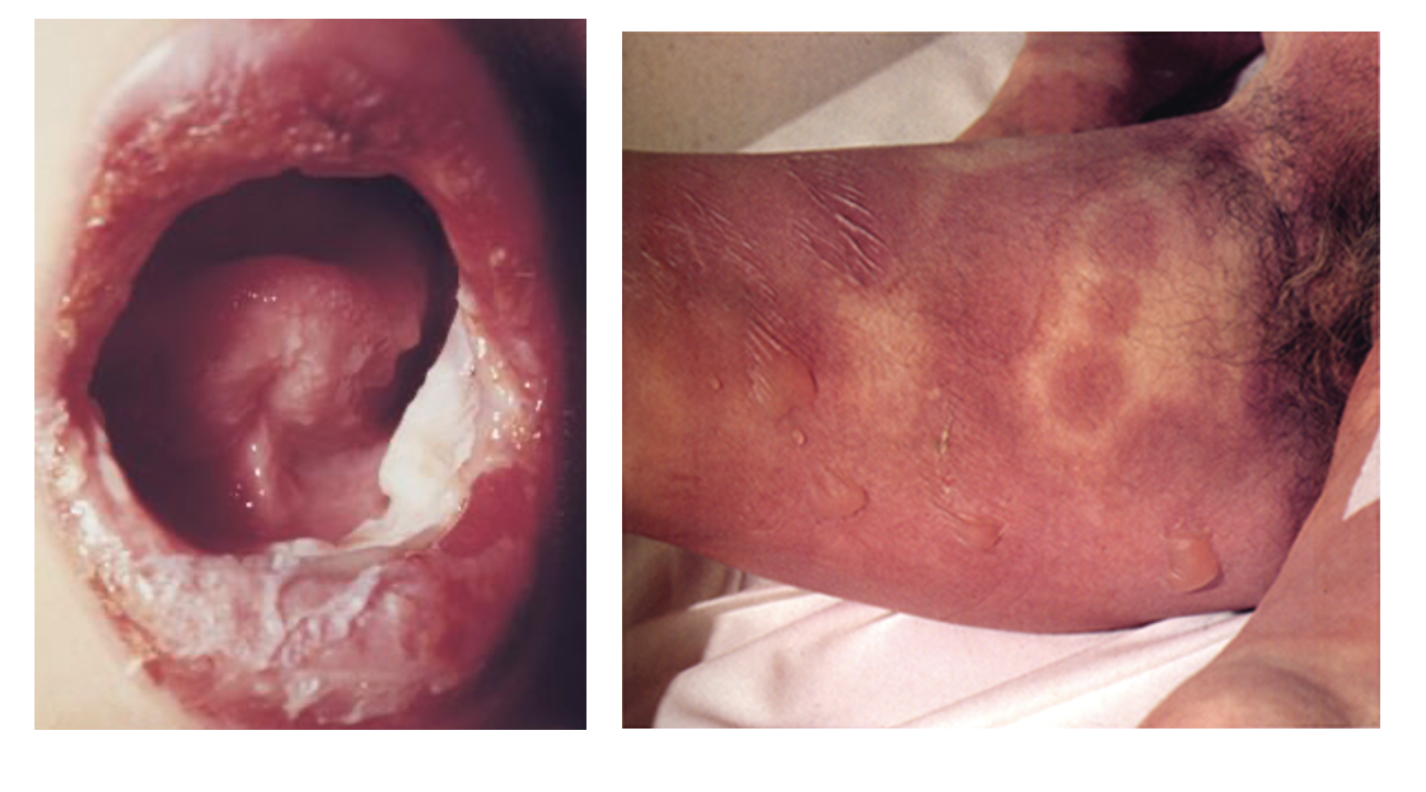
Stevens-Johnson Syndrome
Drug-sensitized cytotoxic CD8+ T cells mediate keratinocyte necrosis
TEN has a mortality rate of approximately 30% that can exceed 50% in elderly or immunosuppressed patients
The severity-of-illness score for TEN (SCORTEN) algorithm facilitates clinical diagnosis and prognostication
SJS is associated with the maintenance of long-lasting tissue-resident memory T-cell responses in the skin that persist after SCAR, necessitating accurate identification and lifelong avoidance of the culprit antibiotic
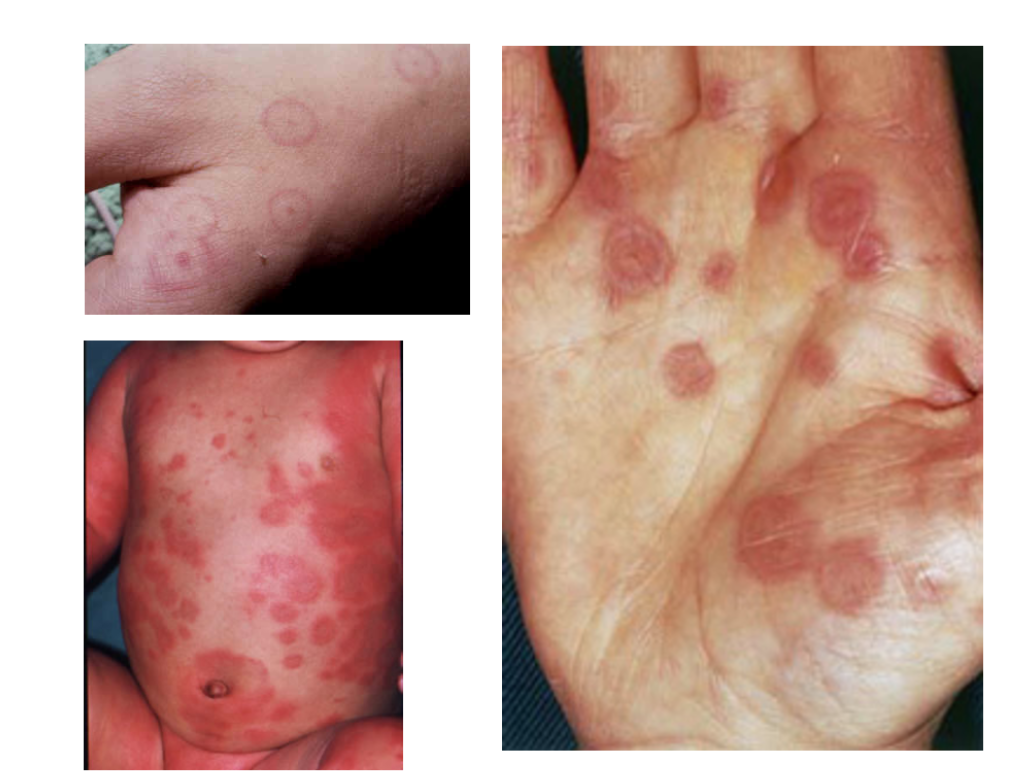
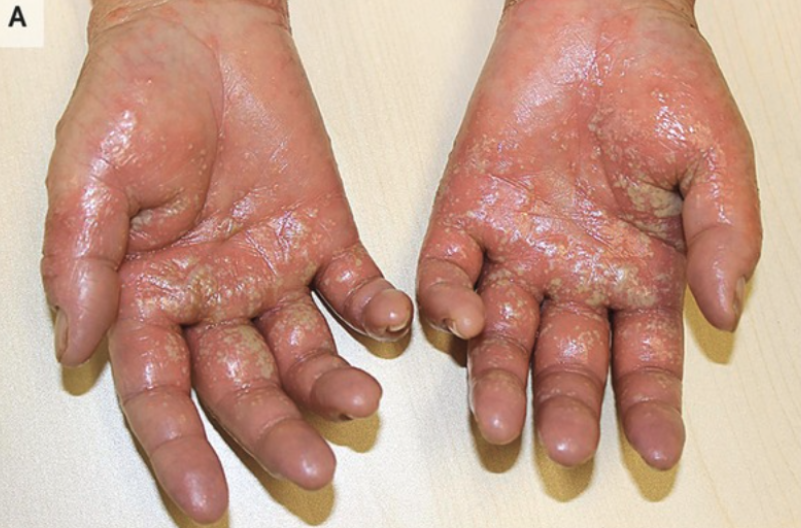
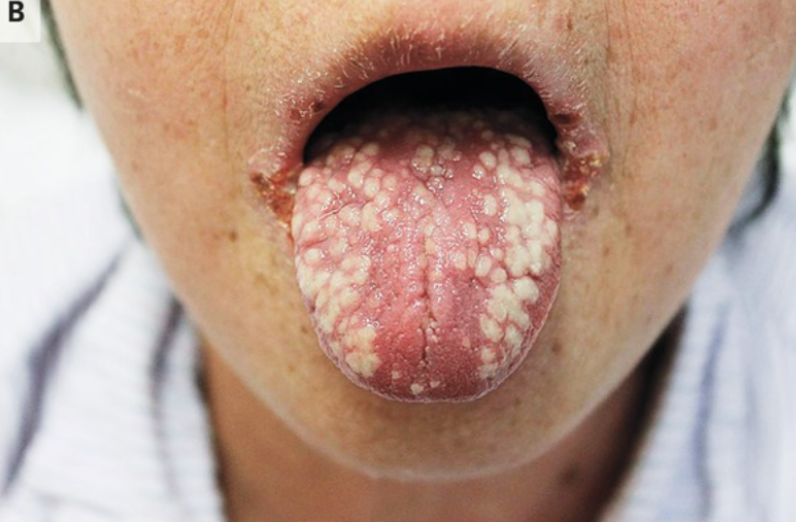
Acute generalized exanthematous pustulosis (AGEP)
AGEP is a drug eruption characterized by an extensive sterile, nonfollicular pustular reaction superimposed on erythematous plaques, with a prominent leukocytosis and neutrophilic dominance
Most cases of antimicrobial-induced AGEP, such as that caused by β-lactams and quinolones, typically cause symptoms within a day of exposure, whereas other drugs take 7 to 14 days of exposure before symptoms.
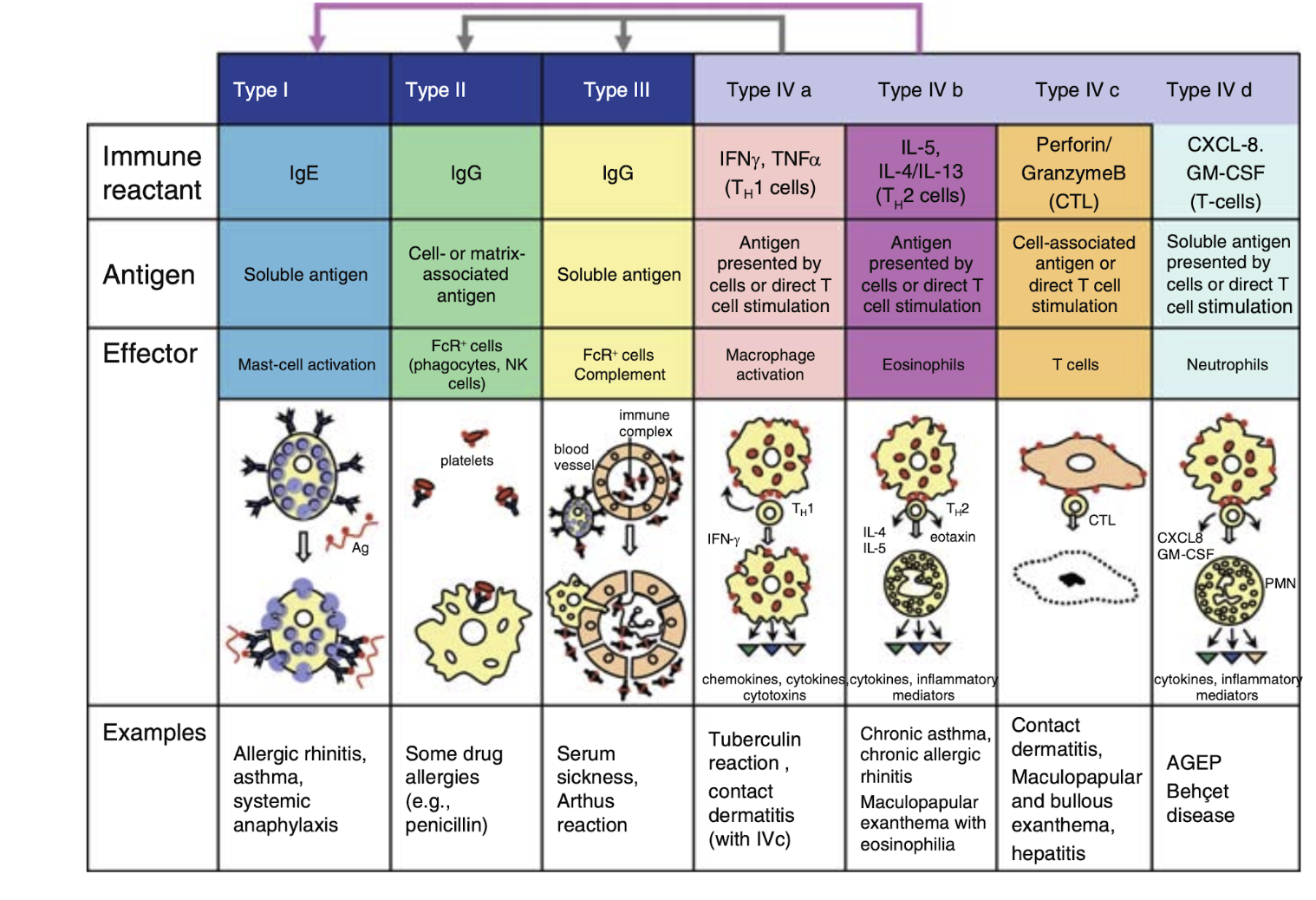
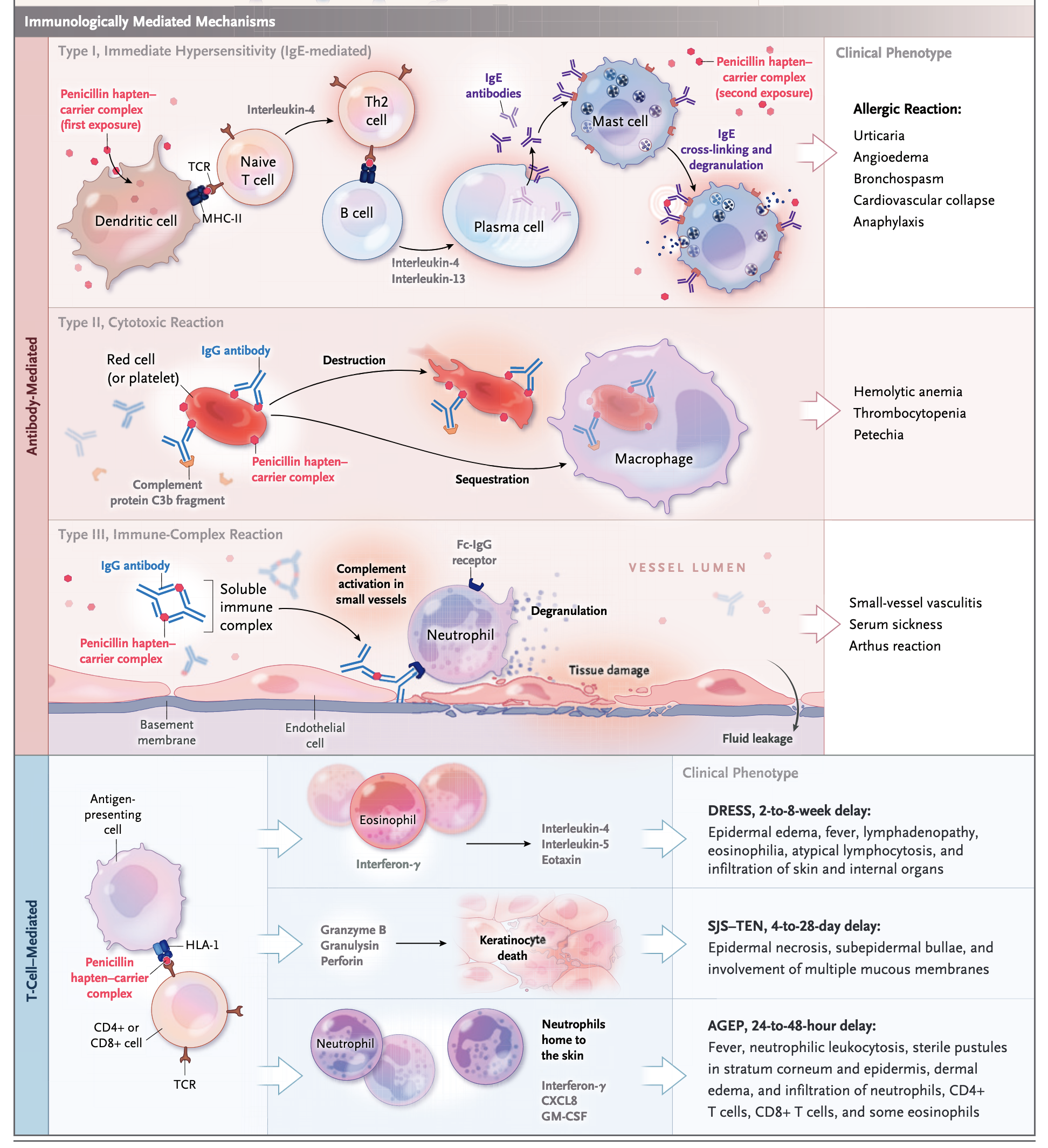
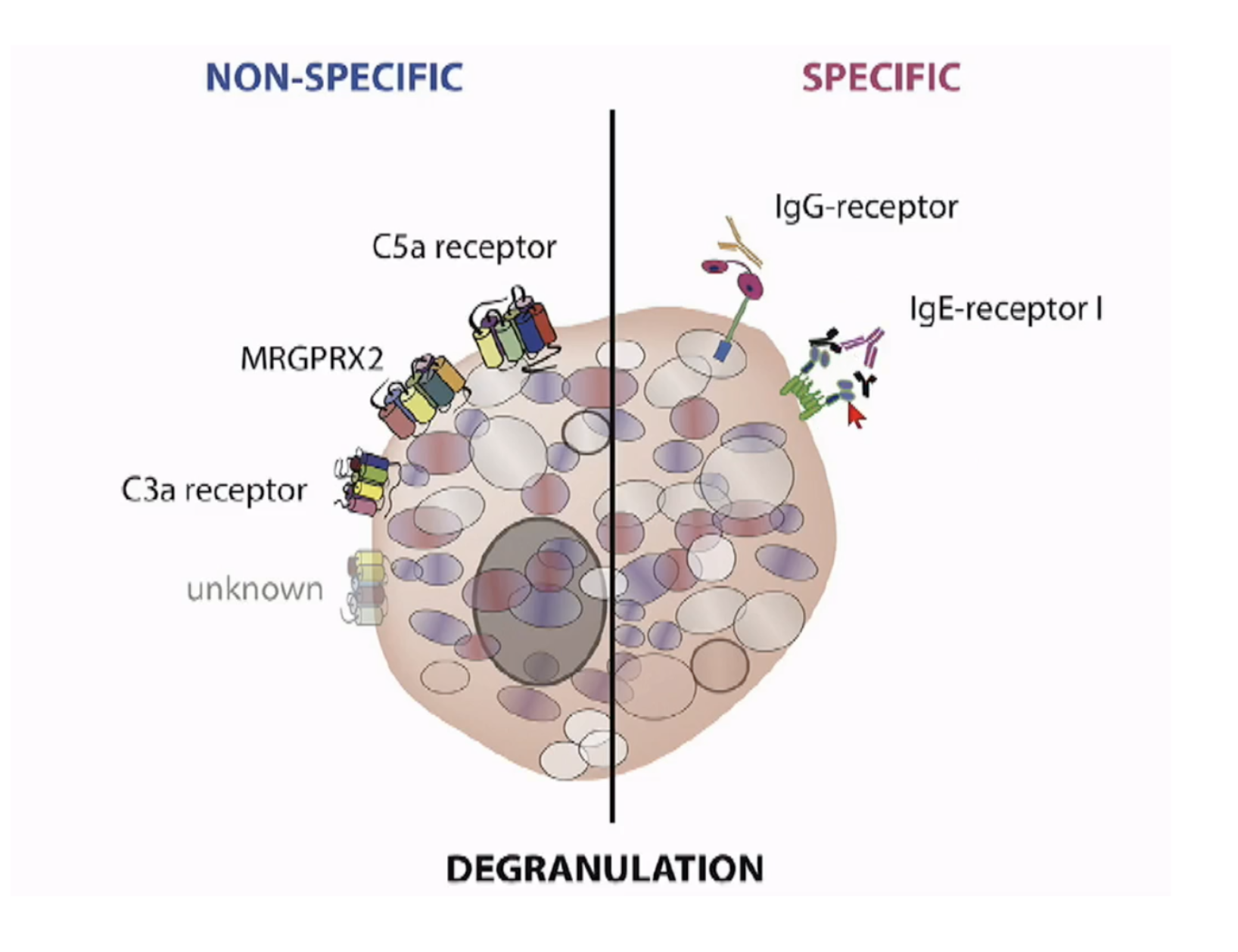
Modified Gell and Coombs Classification
Immunological mechanisms
Penicillin allergy
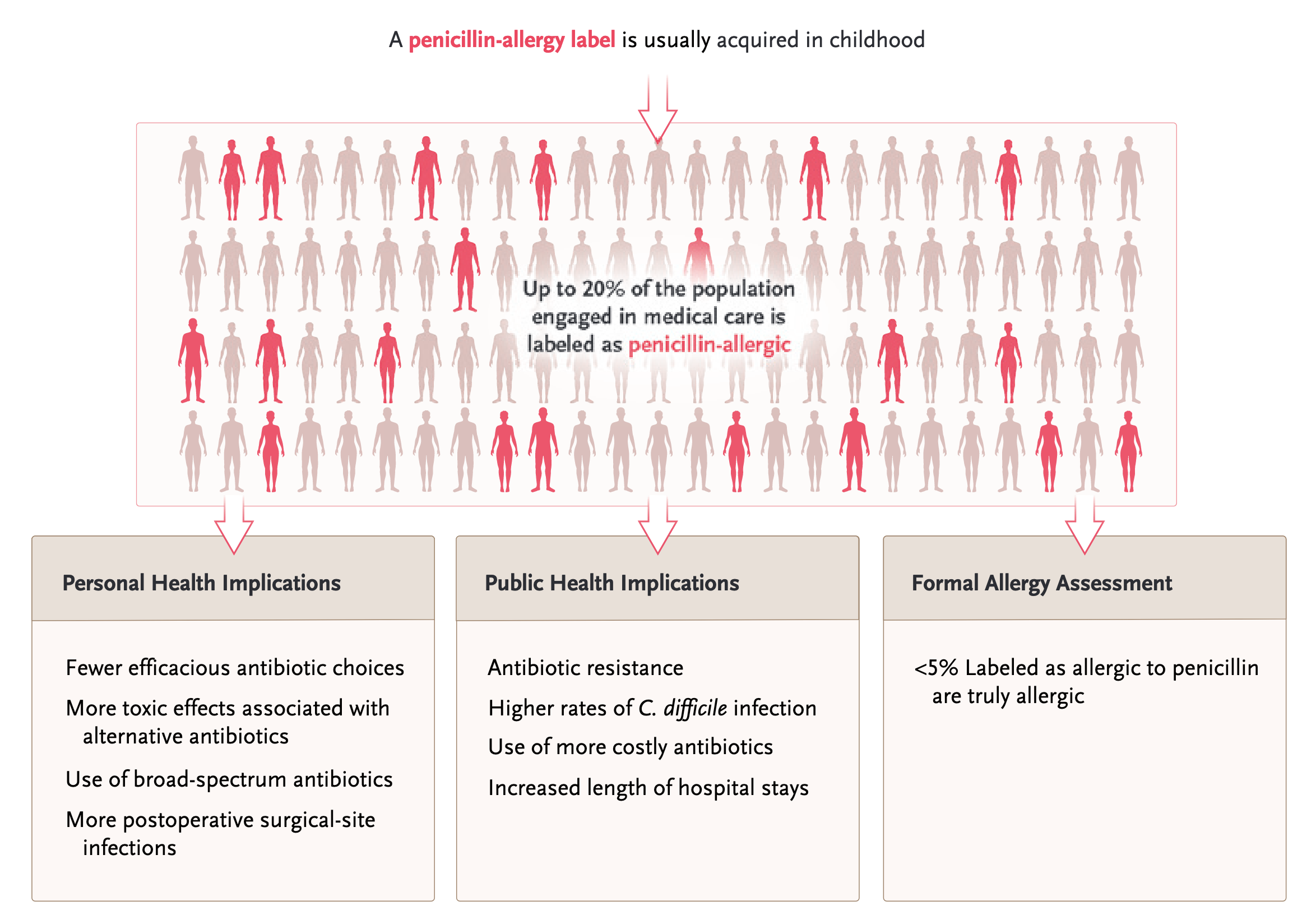
Penicillin allergy epidemiology
10-20% of patients will report a history of an allergy to PCN therapy
However, only 0.5%-2% of all PCN administrations actually result in hypersensitivity reactions, most often rash
- Of these 1% are IgE mediated
The incidence IgE PCN allergies is decreasing, partially due to the reduced use of parenteral PCN, which degradation products in solution may be the primary culprit
Statistics from the UK 1972-2007 oral amoxicillin:
- 1 death after anaphylaxis with oral amoxicillin (35 years and 100 million treatment courses)
Most reports of penicillin allergy describe unknown or cutaneous reaction
Public health implications
So if a patient reports they have a
penicillin allergy…
5% need allergy evaluation
Recent history if true IgE type reaction
Blistering rash
Hemolytic anemia
Nephritis
Hepatitis
Fever and joint pain
Severe cutaneous adverse reaction (SCAR)
95% can tolerate penicillins
- Delayed, benign rash (Type IV reaction) that often does not recur with rechallenge
- True IgE reactions wane over time, with 80% becoming tolerant after 10 years
- Many patients were never allergic, but had other symptoms they though represented a PCN allergy (concurrent viral infection, GI distress)
Top 4 patient penicillin allergy myths
Once you have an penicillin allergy, you have it for life
- Allergy wanes over time, 80% of patients with type I (IgE-mediated reactions) will not have an allergy after a 10 year period
Viral rashes mistaken for antibiotic therapy
E.g., child with a viral exanthematous rash treated with a course of penicillin
Pediatric studies have reported >90% of children who developed rashes on antibiotic therapy do not develop a rash when rechallenged with penicillin again
Adverse effects mistaken by the patient as drug allergy
- E.g. diarrhea, stomach cramps
“I have a family history of penicillin allergy”
- No genetic basis has been identified for penicillin allergies
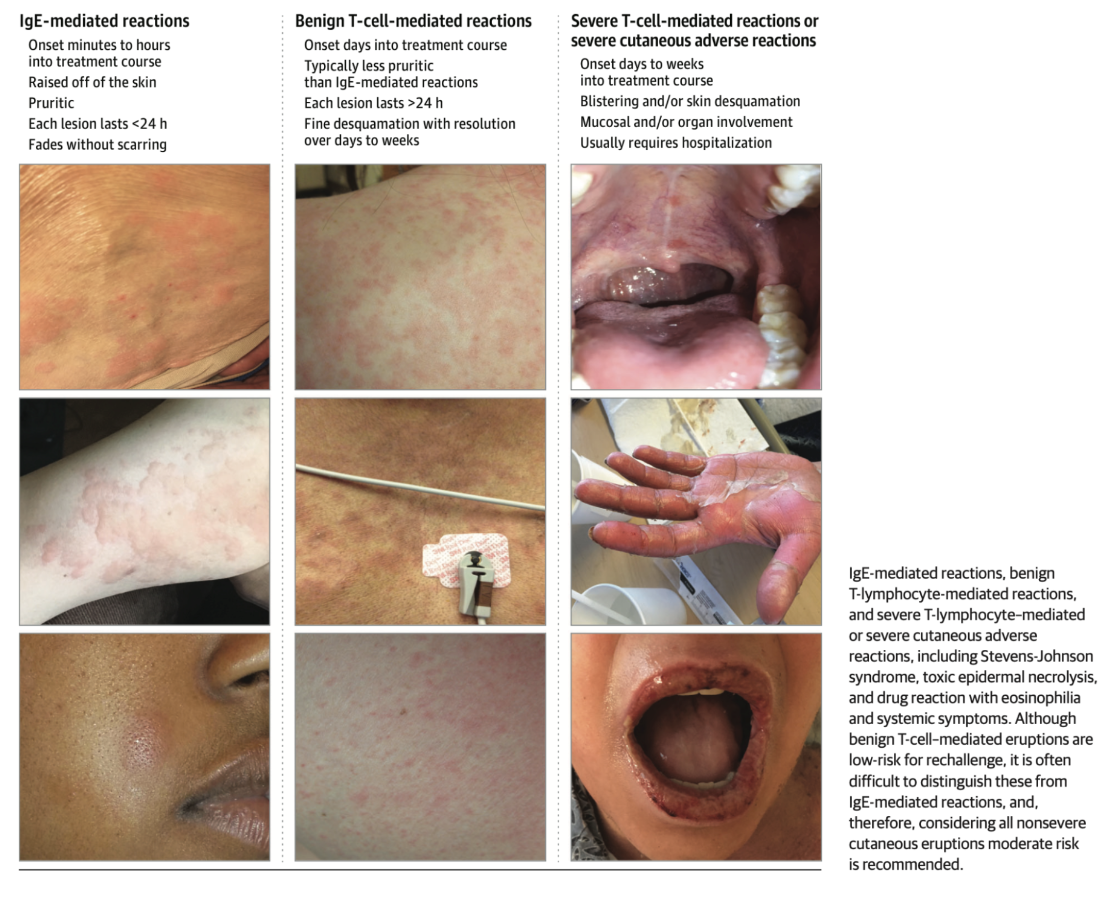
Penicillin allergy history
Timing and clinical presentation of reaction
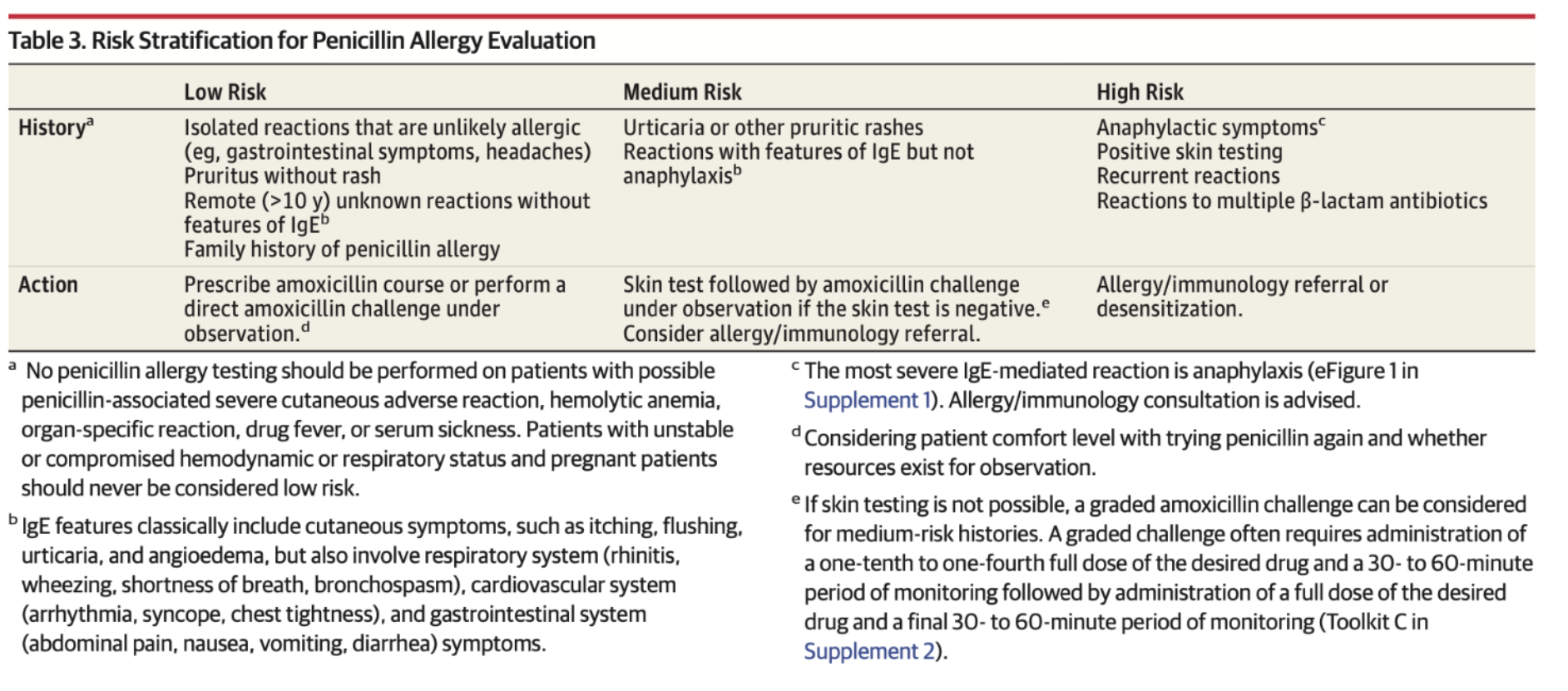
How to assess patient risk
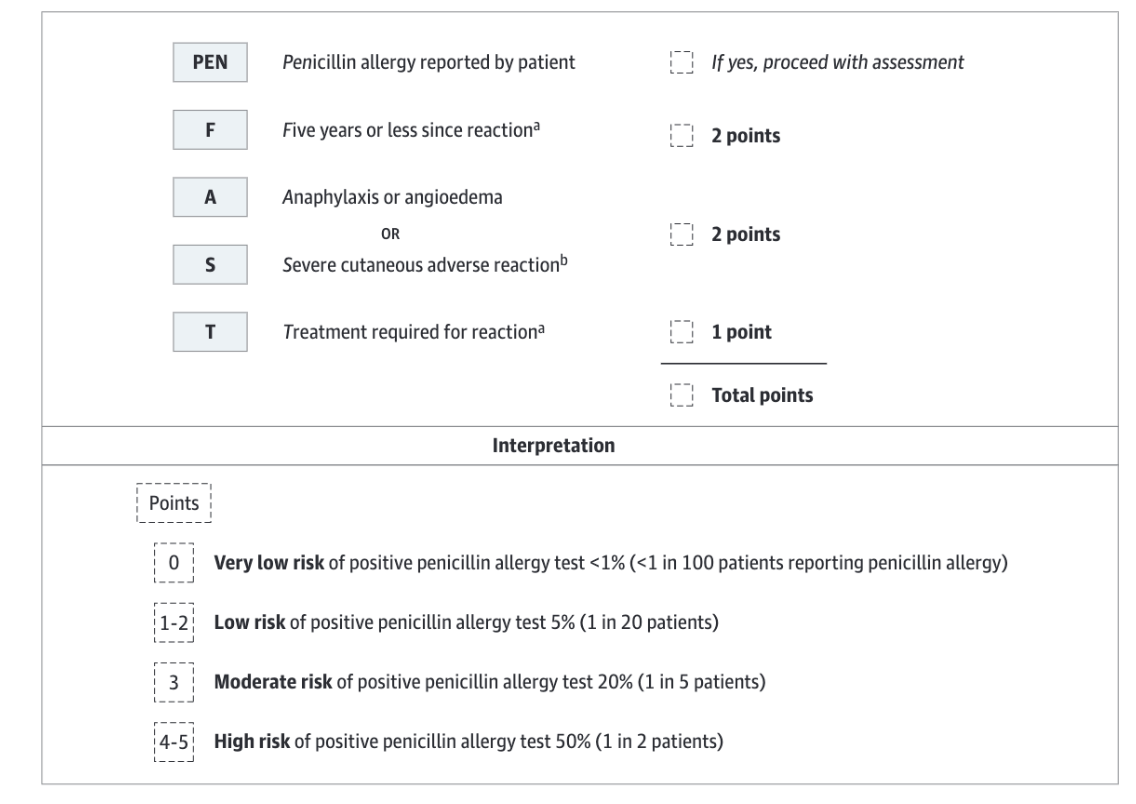
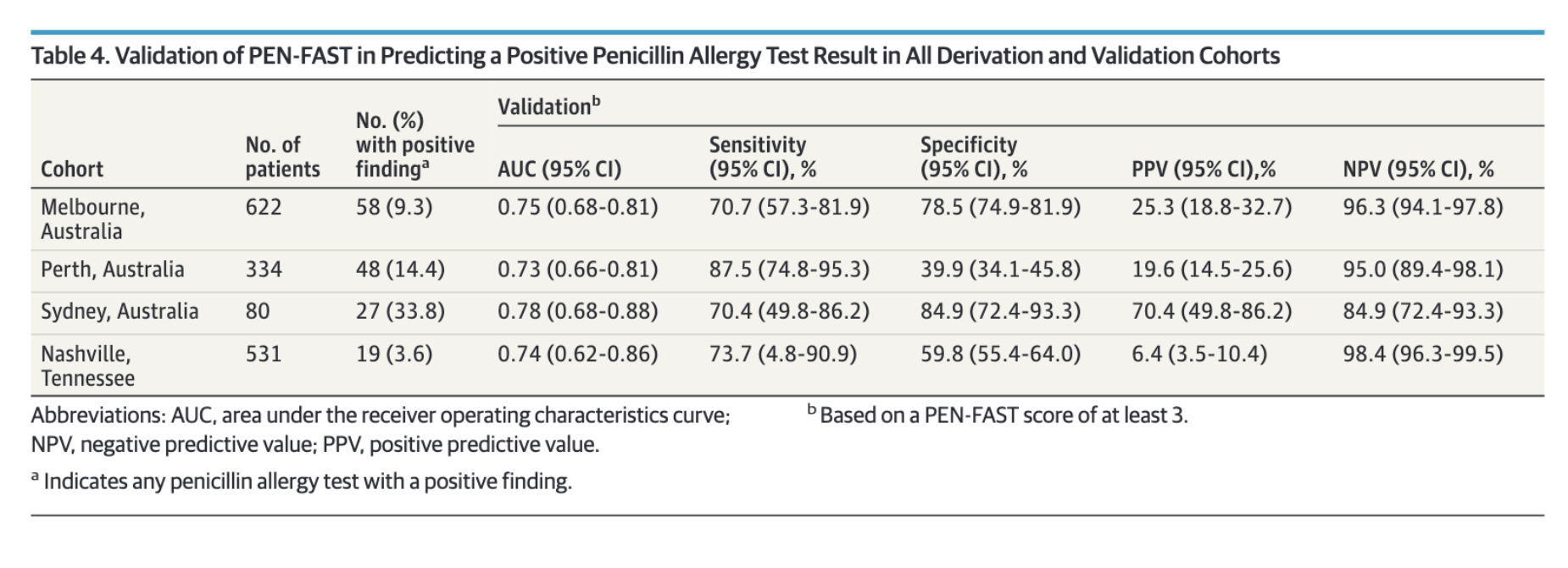
PENFAST score
PENFAST performance
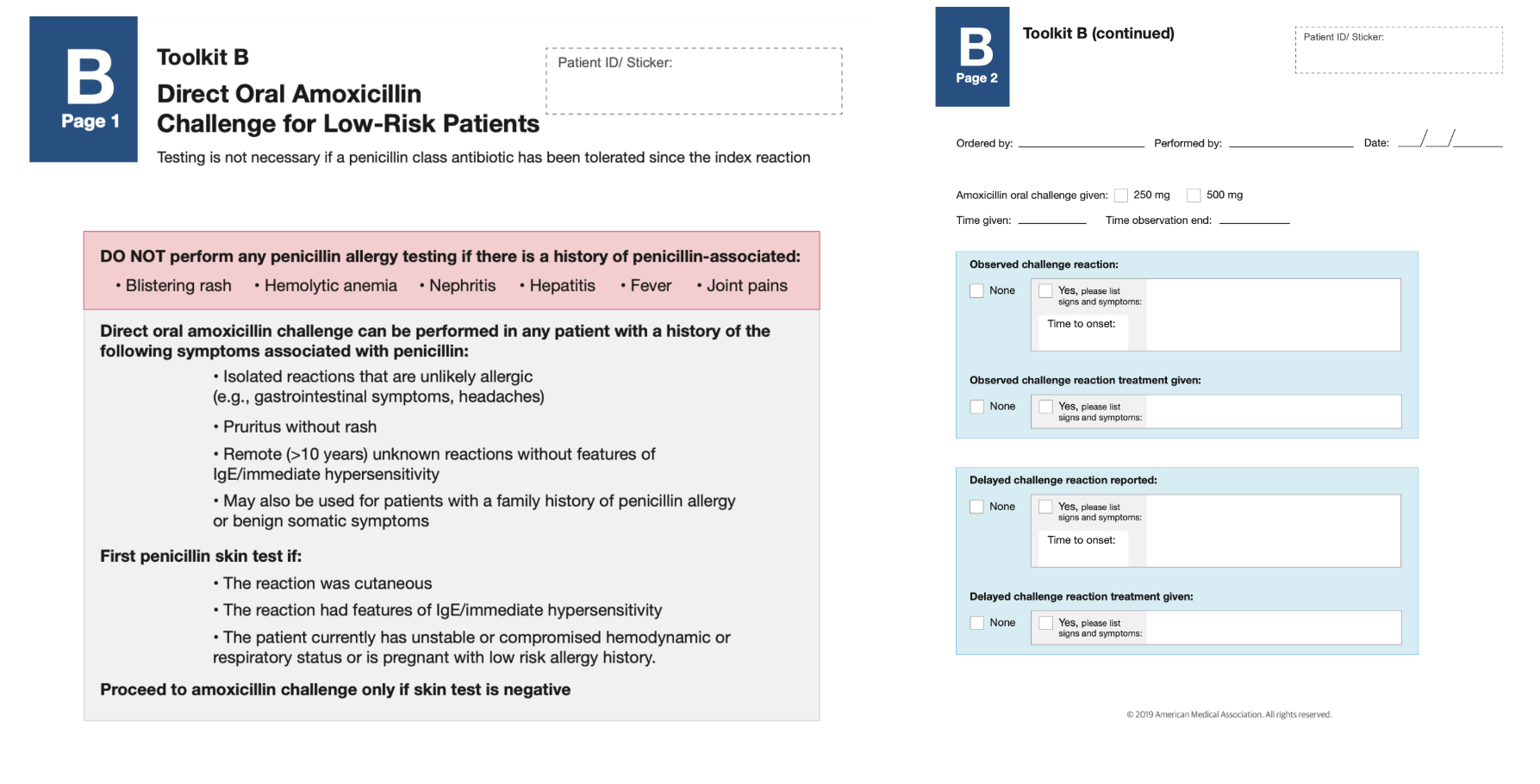
Direct oral amoxicillin challenge
Rationale behind oral amoxicillin challenge
- Only ∼2% of penicillin “allergic” individuals develop an acute hypersensitivity reaction with an oral challenge of a therapeutic dose and 1 hour of observation.
- An additional approximately 2% will have a delayed onset, typically benign, rash within the next 5 days.
- The requirement for viral costimulation of T cells likely explains the low reproducibility of drug exanthems.
- Hence, a direct oral challenge with a single therapeutic dose of amoxicillin is an important step to avoid unnecessary penicillin allergy labels, and indicated in low-risk phenotypes and nonanaphylactic symptoms, such as those with benign delayed-onset rashes without systemic symptoms or SCAR features
Palace study
Two step oral amoxicillin challenge
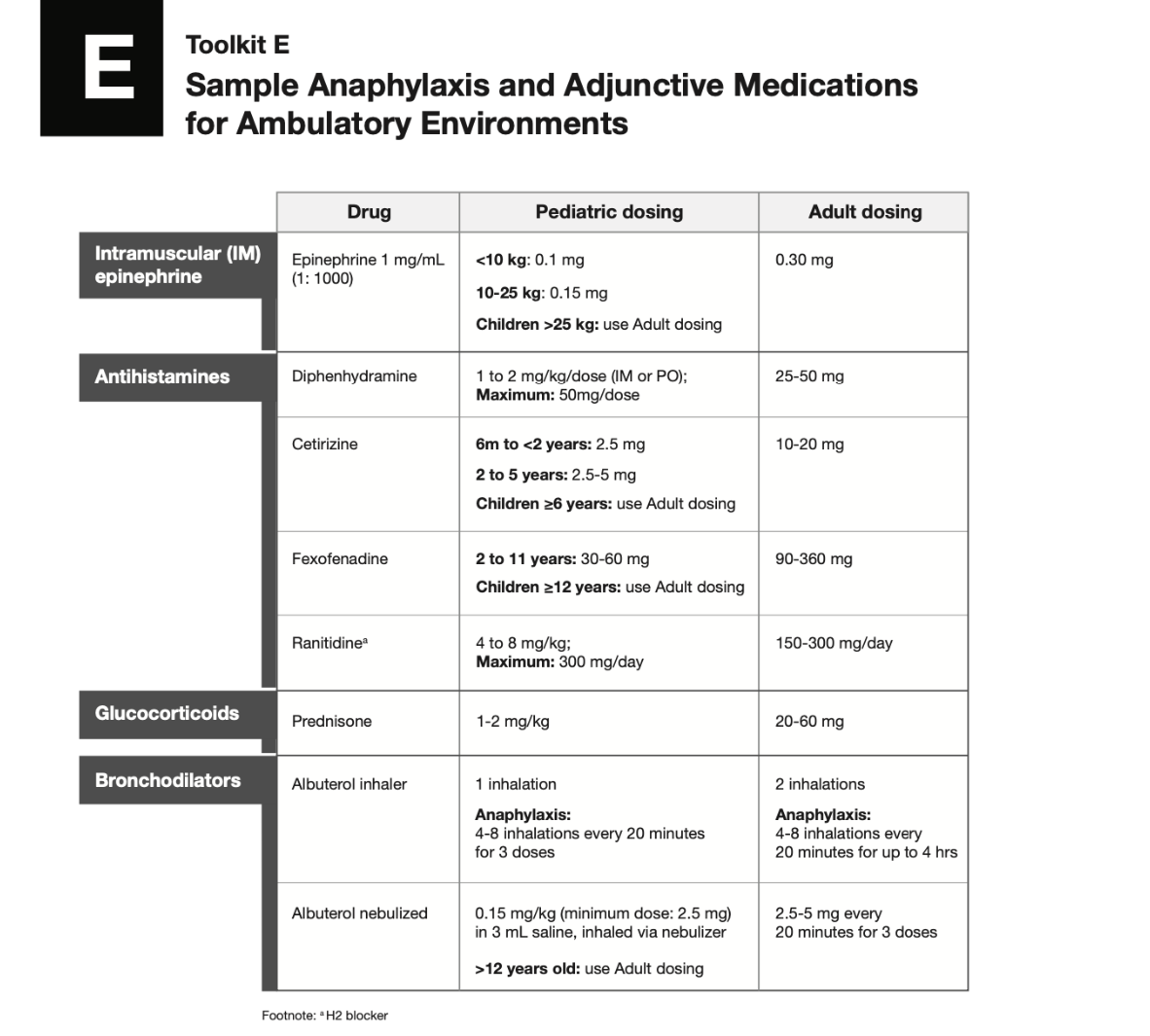
Anaphylaxis medications
How to assess patient risk
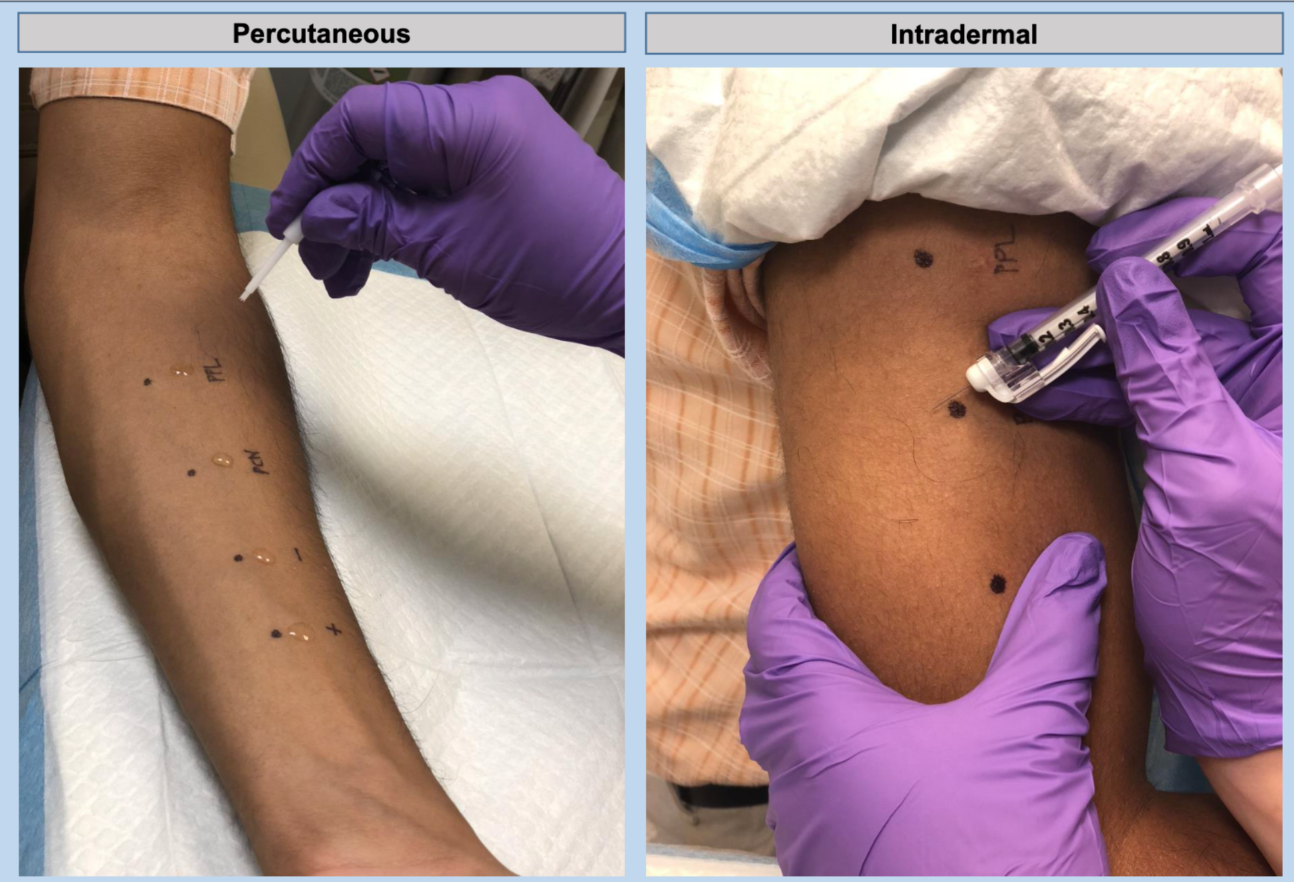
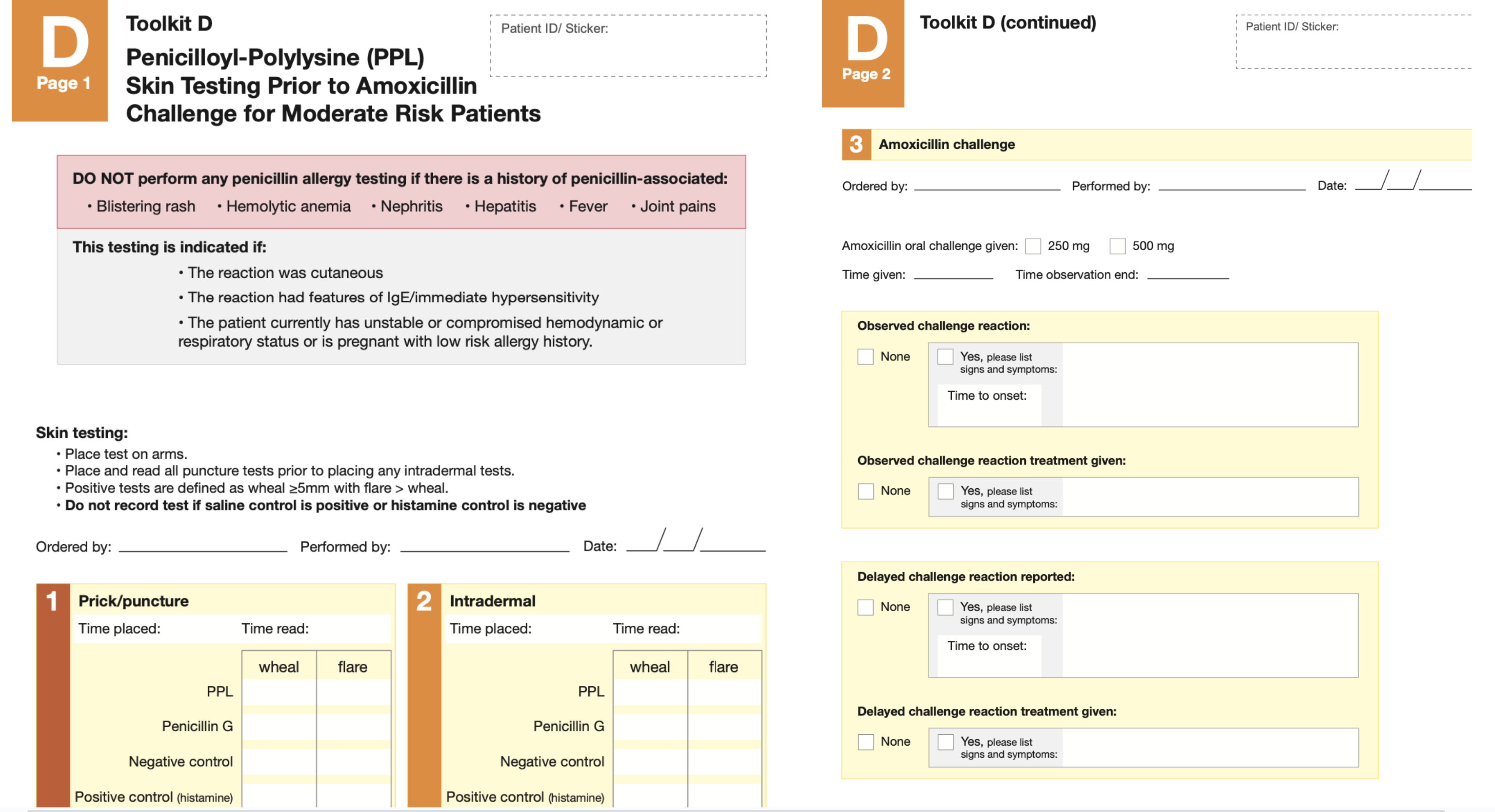
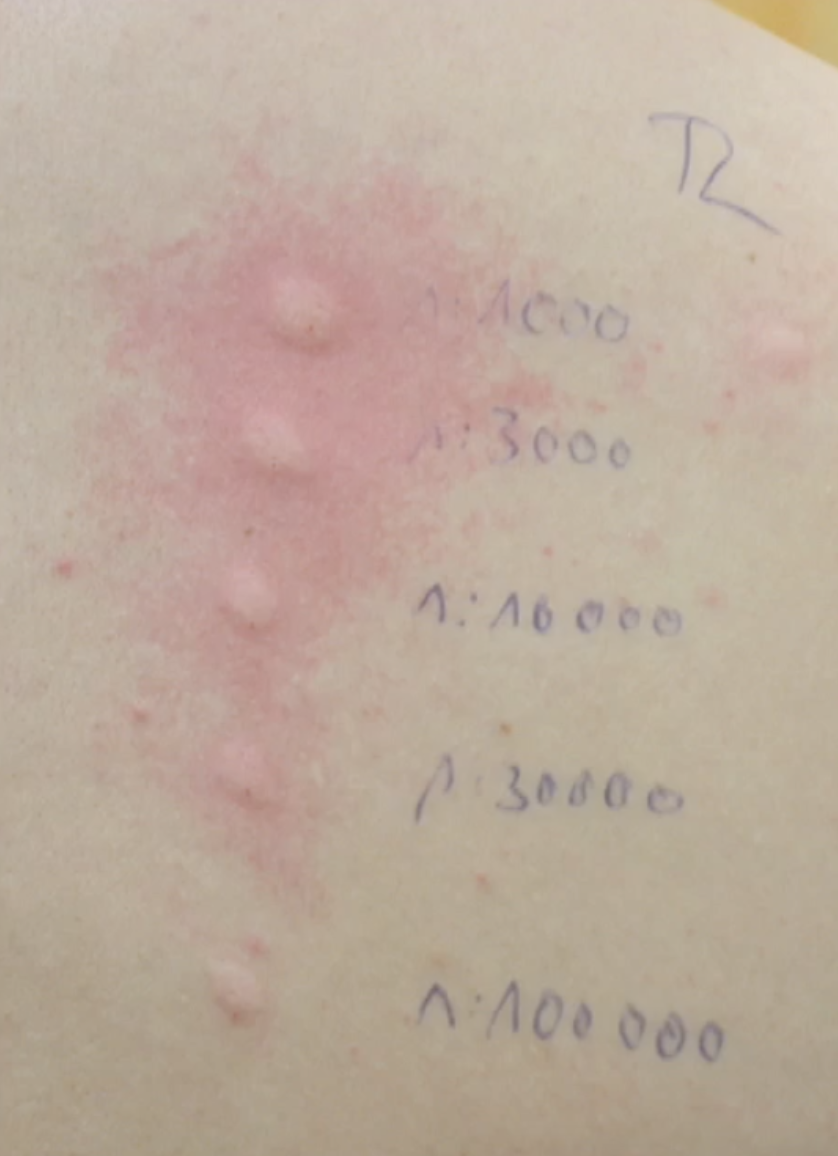
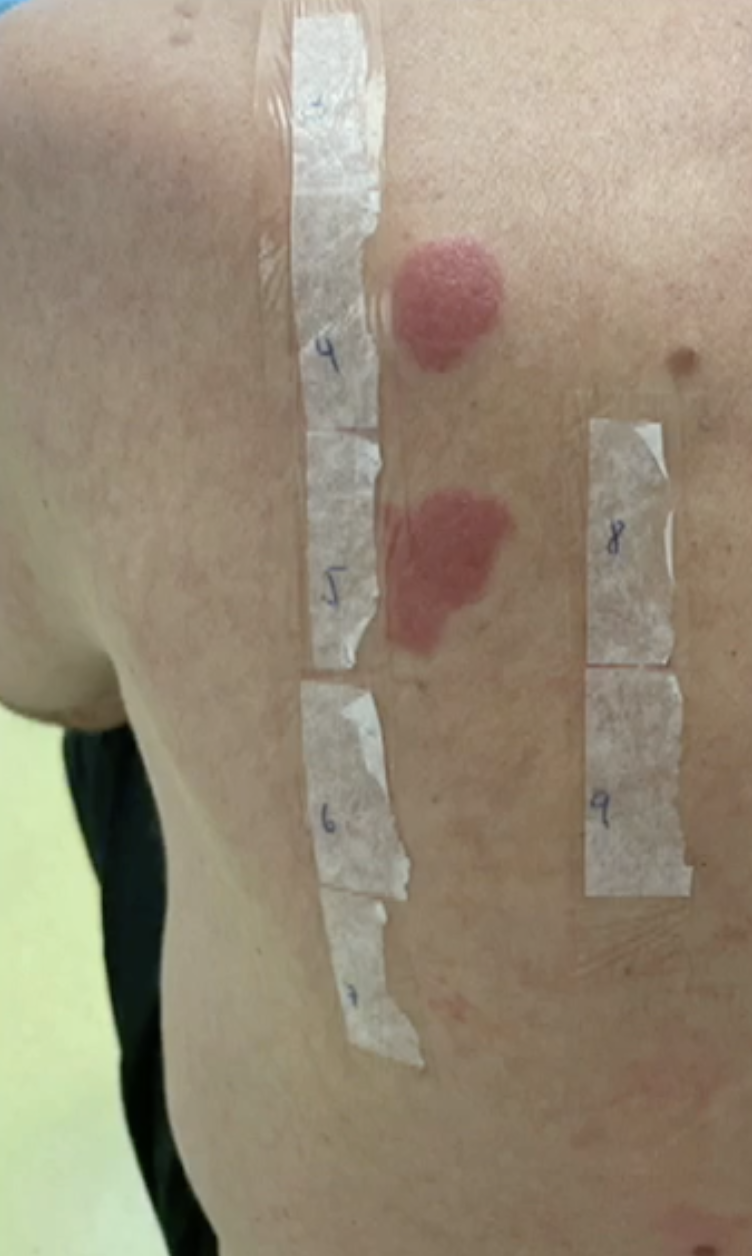
Skin testing
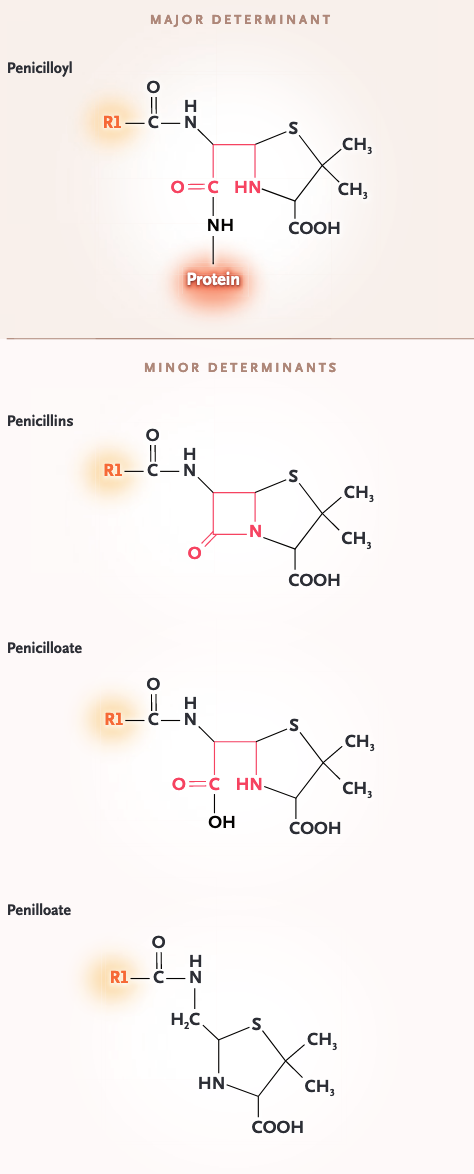
Allergic determinates
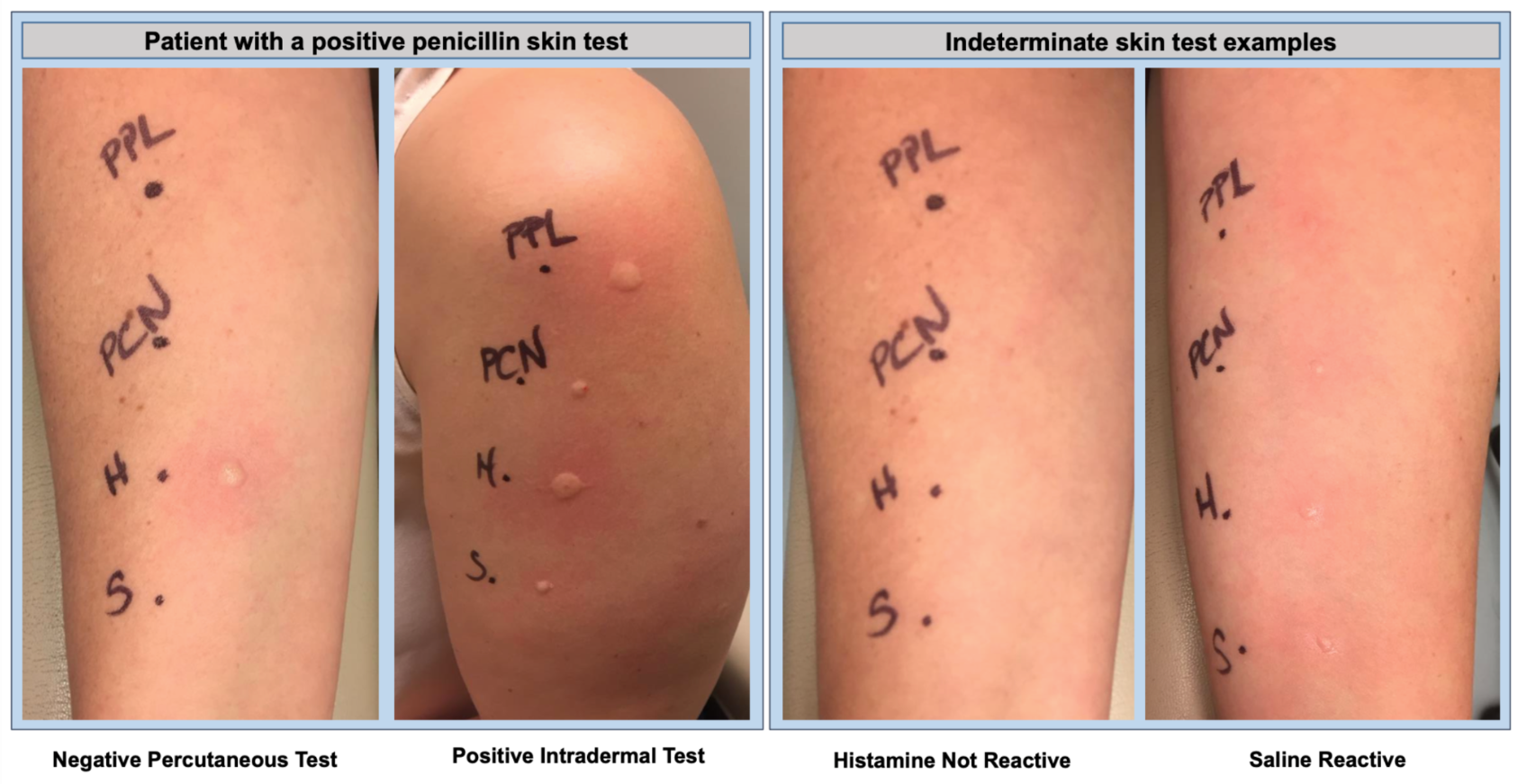
Penicillin skin test assessment
High negative predictive value (NPV) ~95% for IgE-mediated reactions
Poor positive-predictive value- Possible false positive diagnosis if used in patient with low pre-test probability
Amoxicillin is commonly used to challenge after negative penicillin skin tests as it addresses the core β-lactam of penicillin and also side-chain–specific reactions, which may not be detected through skin testing with penicillin itself (NPV 100% if both are negative)
Traditional penicillin skin testing or amoxicillin challenge may be negative in patients with historical reactivity to piperacillin-tazobactam. Skin testing to piperacillin-tazobactam may help to establish this selective sensitization, and these patients are often able to tolerate other penicillins
Penicillin skin testing has no value in delayed reactions, including SJS/TEN, DRESS, and other noncutaneous organ-based reactions
Penicillin skin test assessment
Penicillin skin test assessment
-Medium risk history
Impact “medium risk” patients?
- A negative skin test is associated with a 95% NPV for PCN allergy
- A negative skin test plus negative amoxicillin challenge approaches 100% NPV for PCN allergy
- If skin test is positive, amoxicillin challenge is not considered
- Patient should be referred to an allergy/immunologist or desensitization considered
Desensitization
- Progressive, graded de-granulation of mast cells (histamine release) and internalization of high-affinity IgE receptors by administering graded doses of antibiotic
- Desensitization is contraindicated in patients with a history of a penicillin-induced exfoliative dermatitis, Stevens-Johnson syndrome, or toxic epidermal necrolysis
- Desensitization has no effect on the incidence of non-IgE mediated reactions such as serum sickness, hemolytic anemia, maculopapular rashes, drug fever, hepatitis, or interstitial nephritis
- If the patient does not receive a dose for a period of more than 24 hours, the risk for an immediate IgE-mediated reaction can be restored and repeat desensitization is required if the same drug is to be used again
Sample desensitization protocol
Other desensitization protocols
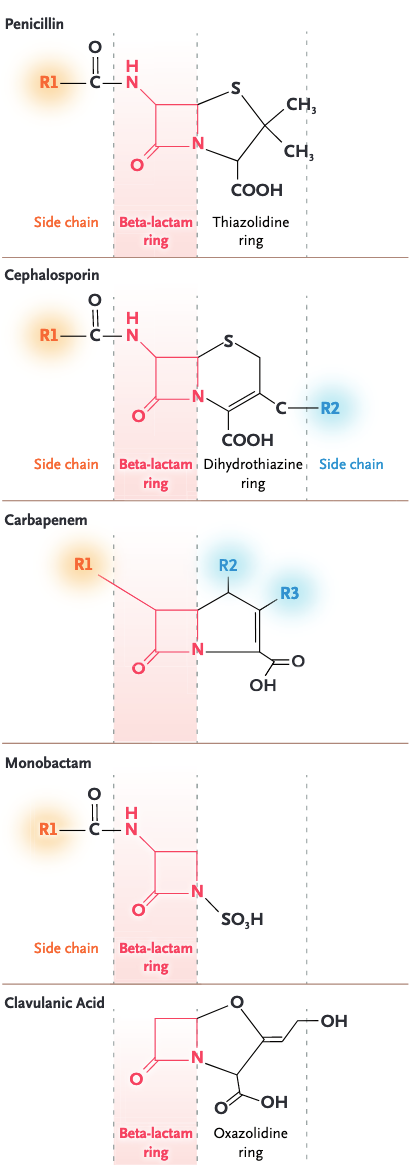
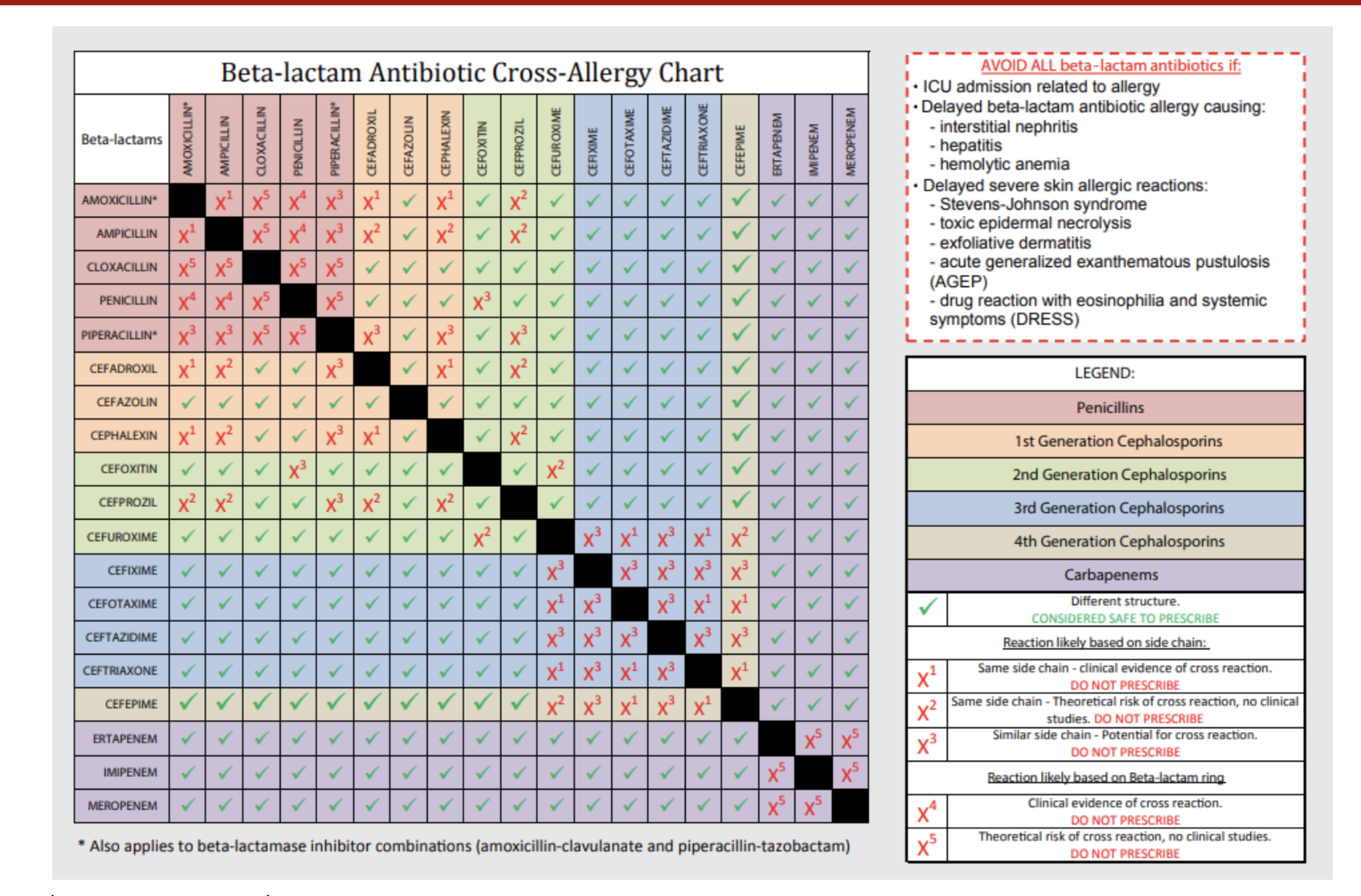
Cross-reactivity: Infectious diseases dogma
- Common teaching: If a patient has a documented PCN allergy, the risk of cross reactions with cephalosporins is 10%
- THIS IS FALSE, the actual cross reactivity is more likely 2%-3%
- A subset of patients with history of anaphylaxis may have cross-reactivity determined by the R1 sidechain of the molecule
- Cefazolin as a unique side chain and and very low risk for cross-reactivity
Risk of cross-reactivity related to cephalosporin sidechain
β-lactams with common side chains
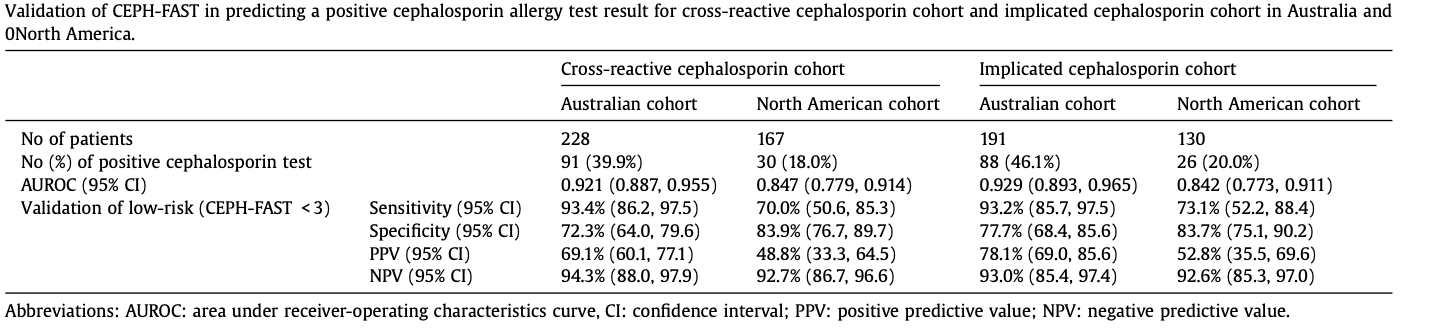
CEPHTEST: Application of PENFAST
score to cephalosporin allergies
What is the cross-reactivity with carbapenems?
- Cross reactivity with penicillin allergy and carbapenems is less than 1%
- No cross reactivity between penicillins and monobactams (aztreonam)
- A graded challenge or test dose can be considered:
- i.e. Infuse 5-10% of dose and observe patient, then progress to full dose if not reaction
- Skin testing (if available) could also rule out allergies
Special populations that should be
considered for testing
- Peri-procedure before elective surgery
- Importance of antibiotic timing/tissue levels at time of incision-less optimal with vancomycin that requires longer infusion
- Pregnancy
- PCN allergy associated with increased risk of cesarean delivery, post-cesarean wound complications, and longer length of stay
- Consider third trimester referral for testing in patients with planned cesarean delivery, group B streptococcus colonization
- Long term care facilities
- Non-beta-lactam based therapies have higher risk for drug interactions
- Higher risk for adverse effects
- Hematology-oncology
- Consider testing before chemotherapy or transplant (onset of of immunosuppression)
- STD clinics
- ICU patients?
Non-penicillin allergies
Similar principles- patient history is key
- Which drugs was the patient taking?
- Dates of intake
- Exact sequence of events
- Underlying disease/Concomitant infections
- Other drug administered at the same time/concomitant infections
- Clinical morphology (at several time points)of rash
- Histology of eosinophilia?
Intradermal testing-Non beta-lactams
- Skin tests for most antimicrobial agents lack high negative predictive values, and skin test positivity is often a function of the time elapsed since the index reaction
- Prick and intradermal tests less well standardized- non-irritating formulations required
- Some antibiotics are irritating even at low concentrations, making testing difficult
- Special cellular activation tests may be available in some centers for some drugs
- e.g., The BAT (flow cytometry) detects the upregulation of activation markers CD63 and CD203c on the surface of basophils after incubation with the implicated drug
Approach to delayed-type
hypersensitivity testing
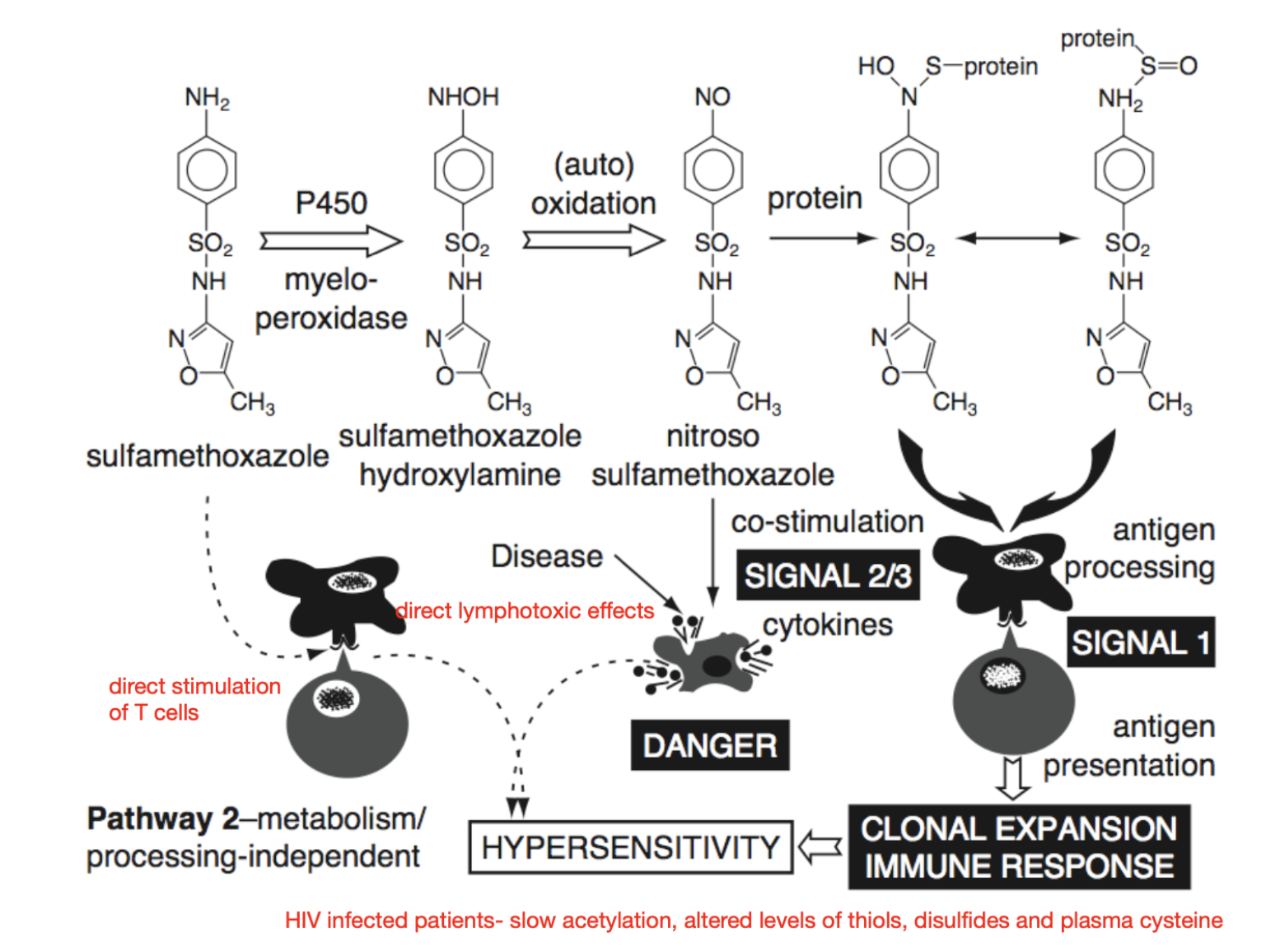
Sulfonamide hypersensitivity
- Incidence 8%
- Primarily cutaneous and GI tract
- Only 3% are considered true hypersensitivity reactions.
- Most common presentations are limited exanthems and fixed drug eruptions
- However…sulfonamides are disproportionately associated with infrequent severe side effects (i.e. TEN, Stevens-Johnson Syndrome)
- Mechanisms IgE-mediated are known to occur, but other poorly understood direct T-cell mediated mechanisms are more likely to be responsible
- Higher incidence of reactions in patients with HIV/AIDS, tuberculosis
Mechanism of sulfonamide hypersensitivity
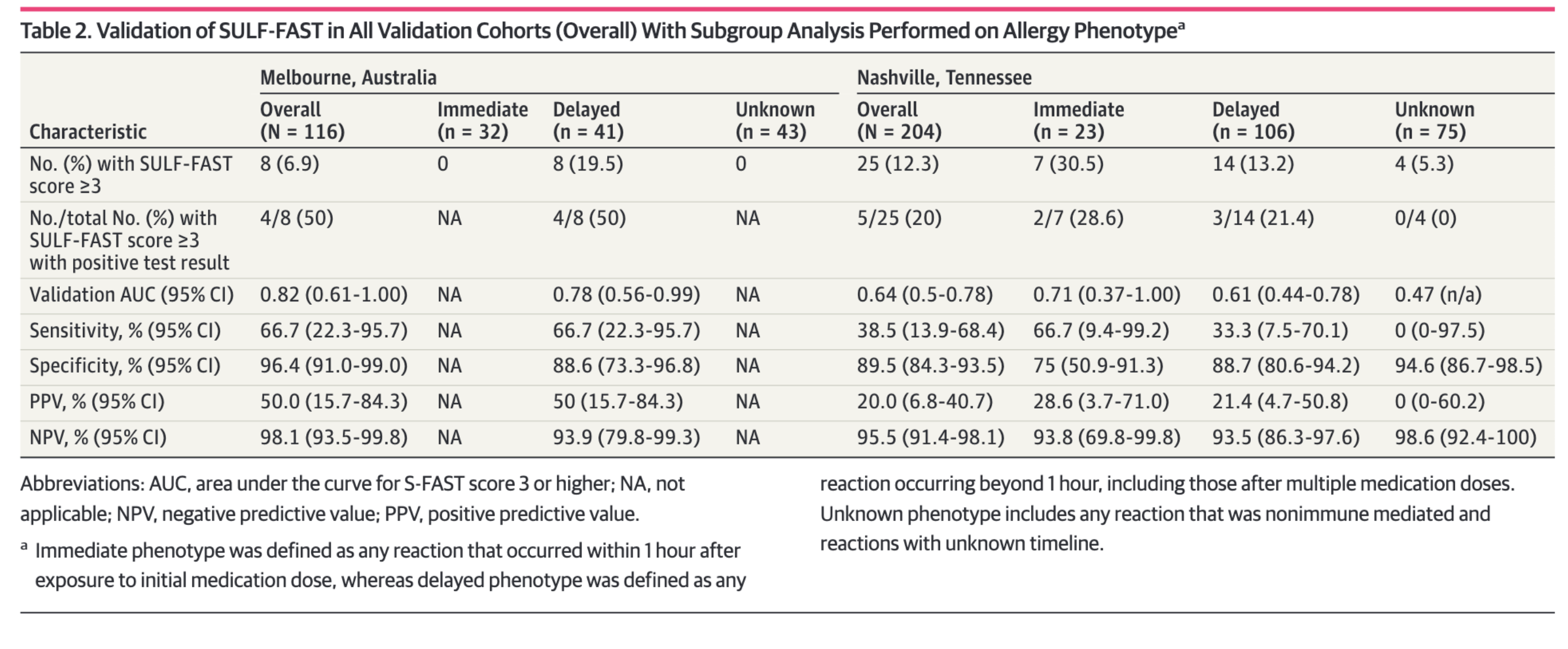
PENFAST score applied to sulfa allergies
Non-sensitizing allergic reactions
Vancomycin infusion reaction
“red man syndrome”
Take home messages
- Penicillin allergies are the most common “contraindication” to antibiotic therapy, but most histories do not represent true allergies
- A systematic approach can be used to evaluate and potentially “de-label” patients with penicillin allergy
- Cross-reactivity rates are low with current cephalosporins and carbapenems, but can alternatively be addressed through antibiotic challenges and skin testing
- Desensitization can be attempted in specific cases when a particular antibiotic is needed for IgE-mediated reactions
- Some antibiotics cause non-immune-related hypersensitivity reactions that can be managed by slowing infusions and administering antihistamines