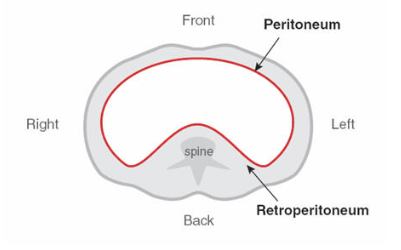
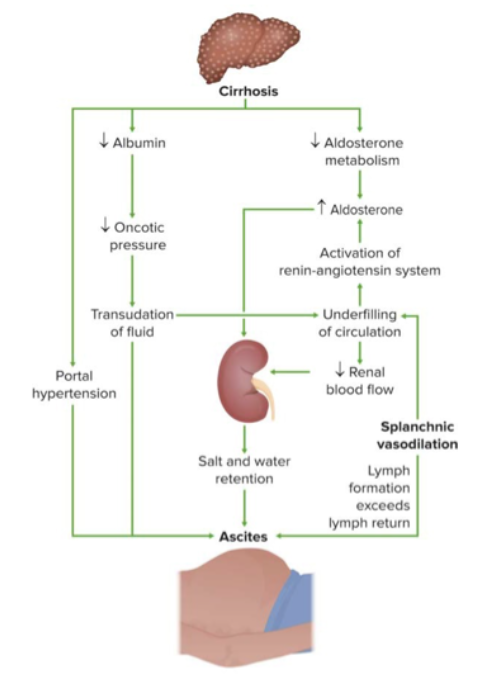
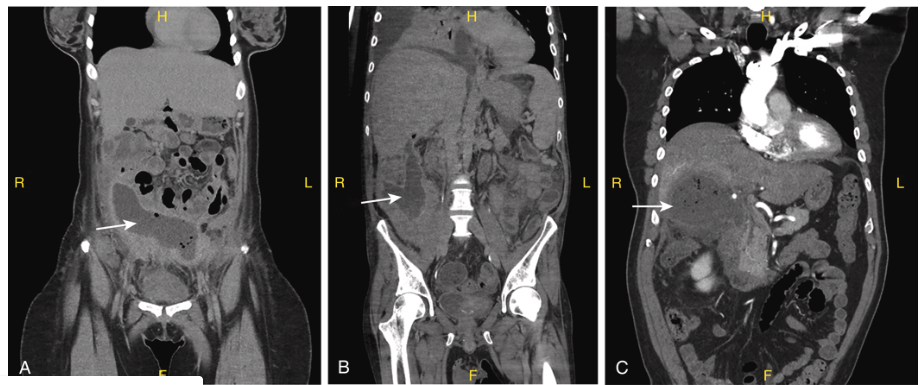
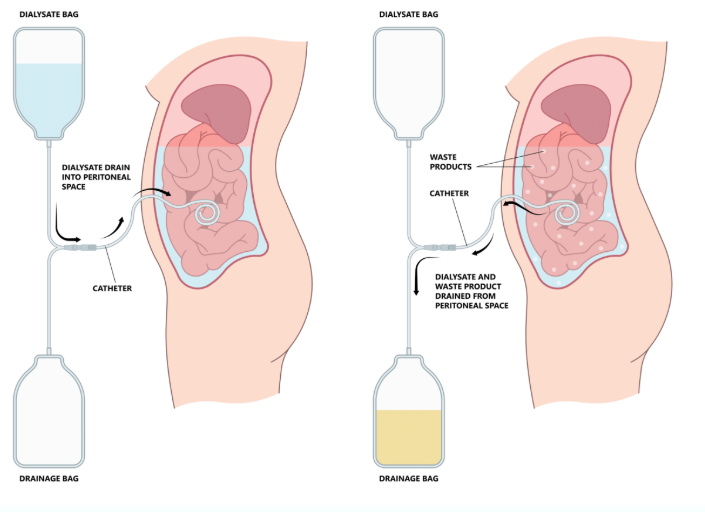
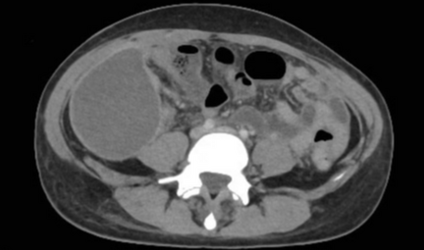
Primary vs. secondary vs. tertiary
| Feature | Primary | Secondary | Tertiary |
|---|---|---|---|
| Intraabdominal source | None | Present (perforation, ischemia) |
Persistent after source control |
| Frequency | ~1% of peritonitis cases | 80–90% of cases | Less common |
| Typical organisms | Monomicrobial gram-negative | Polymicrobial aerobic/anaerobic | MDR, gram-negative, low virulence |
| Mortality | 5–20% | 10–40% | 30–64% |