Immunosuppression: An overview of infection risk
2026-03-01
Immununosuppression:
An Overview of Infection Risk
Prof. Russell E. Lewis
Department of Molecular Medicine
University of Padua
russelledward.lewis@unipd.it
https://github.com/Russlewisbo
slides available at: www.padovaid.com
 |
Introduction & Epidemiology
The growing population of immunocompromised hosts
- Estimated 5-6% of the Italian population is immunocompromised (Martinson and Lapham, 2024)
- 1% of children fall into cohort of immunocompromised (VERDI project)
- 2.8% meet criteria for drug-induced immunosuppression (Wallace et al., 2021)
What causes immunocompromise?
Major categories:
- Active treatment (chemotherapy) for malignancies
- Solid organ transplant (SOT)
- Hematopoietic cell transplant (HCT)
- CAR-T cell therapy
- Primary immunodeficiency
- Advanced HIV infection
- High-dose corticosteroids & biologics
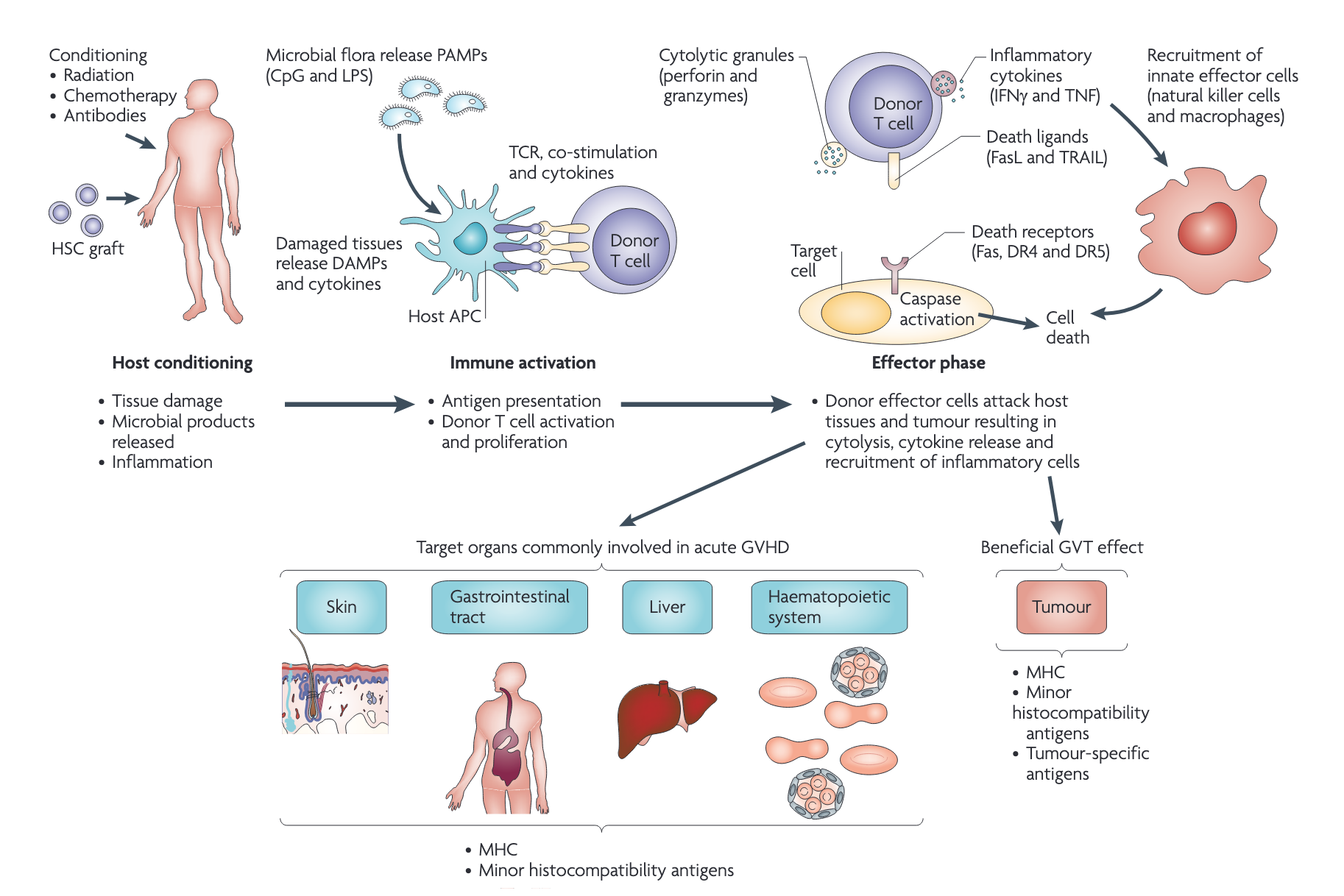
The net state of immunosuppression
Defining net state of immunosuppression
“Father” of transplant infectious diseases”
“Composite of host factors, underlying disease, treatment, and other factors contributing to infection risk”
Components of the “net immunosuppressed state”
Host Factors
- Advanced age
- Malnutrition
- Diabetes
- Organ dysfunction
- Hypogammaglobulinemia
Treatment Factors
- Immunosuppressive drugs
- Chemotherapy
- Radiation
- Surgery/hardware
- Duration of therapy
Components (continued)
Underlying Disease
- Autoimmune disease
- Malignancy type
- Organ failure stage
Infectious Factors
- HIV, CMV, EBV status
- Microbiome alterations
- Prior infections
SOT Recipients
- 6% died from infection within first year (Swiss cohort) (Delden et al., 2020)
- 55% had infections in first year (German renal cohort) (Sommerer et al., 2022)
- Half occurred in first 3 months
- Bacteria: 66%, Viruses: 29%, Fungi: 5%
Hematopoetic stem cell transplantation (HSCT) recipients
| Type | Stem Cell Source | Donor | Immunosuppression |
|---|---|---|---|
| Autologous | Peripheral blood, Bone marrow | Self | Moderate; no GvHD prophylaxis required; recovery within weeks |
| Allogeneic — matched related | Peripheral blood, Bone marrow, Umbilical cord blood | HLA-matched sibling or family member | Severe; prolonged due to GvHD prophylaxis and risk of GvHD |
| Allogeneic — matched unrelated (MUD) | Peripheral blood, Bone marrow, Umbilical cord blood | HLA-matched unrelated donor (registry) | Very severe; higher GvHD risk than matched related; intensive prophylaxis |
| Allogeneic — haploidentical | Peripheral blood, Bone marrow | Half-matched family member (parent, child, sibling) | Very severe; requires intensive T-cell depletion or post-transplant cyclophosphamide |
| Allogeneic — umbilical cord blood | Umbilical cord blood | Unrelated cord blood unit | Very severe; delayed immune reconstitution due to low cell dose |
Allogeneic hematopoetic stem cell
transplantation (HSCT) recipients
Timeline of Infection Risk
What is CAR-T therapy?
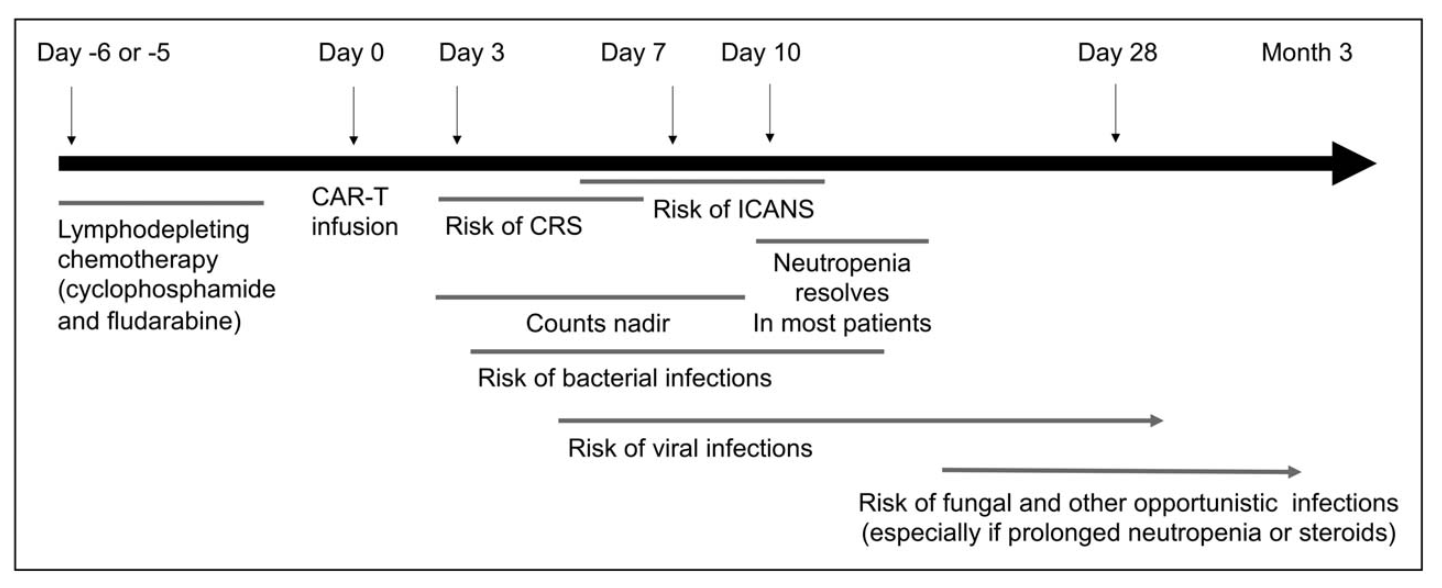
Immunosuppression timeline with CAR-T
CAR-T cell therapy
- Patients are profoundly immunosuppressed
- Up to 1/3 suffer serious bacterial infection in first 30 days (Stewart and Henden, 2021)
- Cytokine release syndrome complicates assessment
- Prolonged B-cell aplasia → hypogammaglobulinemia
Measuring Immunosuppression
Available Markers
Useful in HIV:
- CD4 count
- CD4 percentage
- CD4/CD8 ratio
General markers:
- Neutrophil count
- Lymphocyte count
- Immunoglobulin levels
Emerging biomarkers
- Viral reactivation (EBV, CMV, TTV, BK) → correlates with immunosuppression
- QuantiFERON Monitor → may identify over-immunosuppression
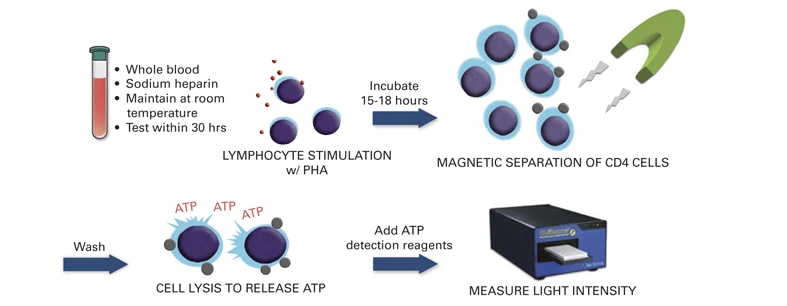
- ImmuKnow assay → correlates with infection/rejection risk
- Traditional markers (ESR, CRP, procalcitonin) → NOT predictive
Sources of Infection
Community-acquired pathogens
- Most common infections mimic community pathogens
- Immunocompromised patients are often “sentinel cases” in outbreaks
- Respiratory viruses, GI pathogens
- Norovirus, C. difficile
Healthcare-associated pathogens
- Increased risk of MDR organisms due to frequent healthcare contact
- Catheter-related infections
- Pneumonia
- UTI
Reactivation of latent infections
Donor-derived infections
- Organ transplant
- Stem cell transplant
- Blood products
- Usually within first 6 months
- Most common infections:
- Cytomegalovirus
- Epstein-Barr virus (post-transplant lymphoproliferative disease)
- Herpes simplex and varicella zoster
- Hepatitis B,C
- HIV
- Bacterial infections
- Fungal infections (Candida, Aspergillus)
Components of Host Defense
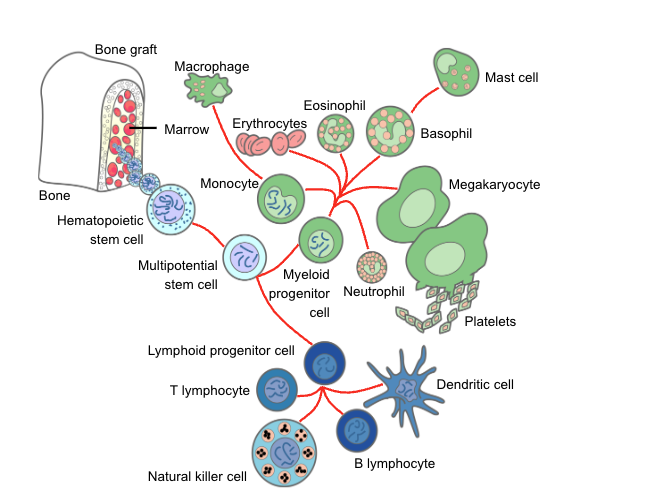
Overview of immune system
Innate Immunity
- Granulocytes
- Monocytes/Macrophages
- NK cells
- Complement
- Physical barriers
Acquired Immunity
- Cellular (T cells)
- Humoral (B cells)
- Antibody production
Granulocytes (neutrophils)
Corticosteroid effects on neutrophils
Paradoxical effects:
- ↑ Granulocytopoiesis (apparent benefit)
- BUT: ↓ Accumulation at infection site
- ↓ Adherent capacity
- ↓ Chemotaxis
- ↓ Phagocytosis
- ↓ Intracellular killing
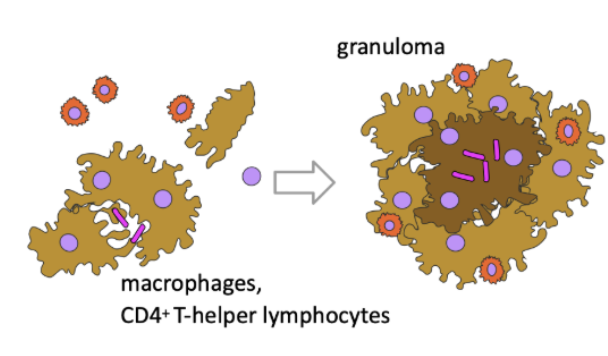
Monocytes & macrophages
- Monocytopenia parallels neutropenia
- Macrophage activation requires T-cell cytokines (IFN-γ)
- Explains cellular immunodeficiency susceptibility
- Targeted therapies can have unexpected effects
NK cells and platelets
NK Cells:
- Respond to viruses and malignancy
- CD56 receptor → Aspergillus recognition
- Dysfunction contributes to fungal susceptibility
Platelets:
Increasingly recognized immune role
Thrombocytopenia → independent bacteremia risk
Protection against yeast and molds
Cellular Immunity
Specific cell-mediated immune defects:
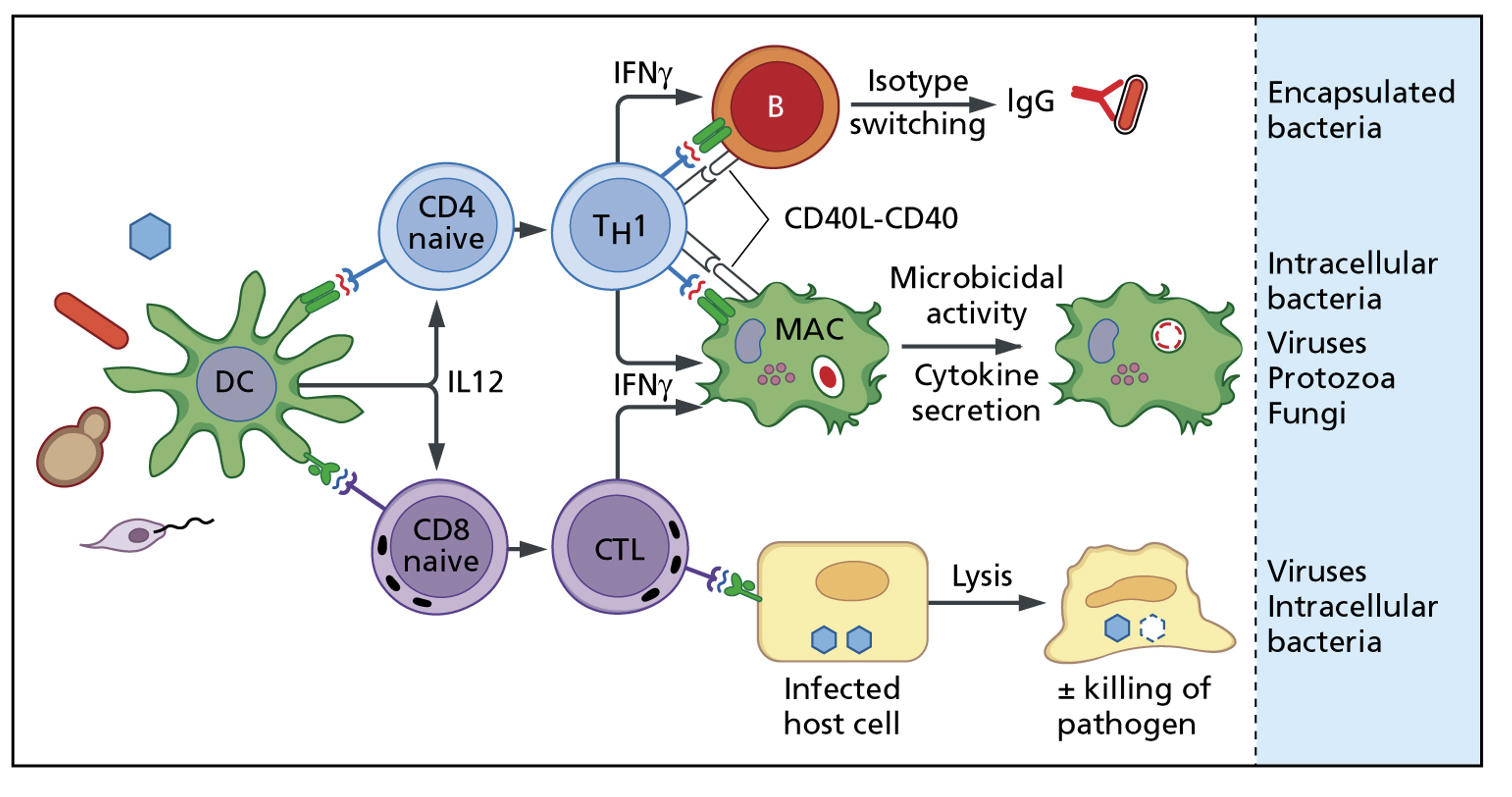
Th1 and CTL-driven immunity
Specific cell-mediated immune defects:
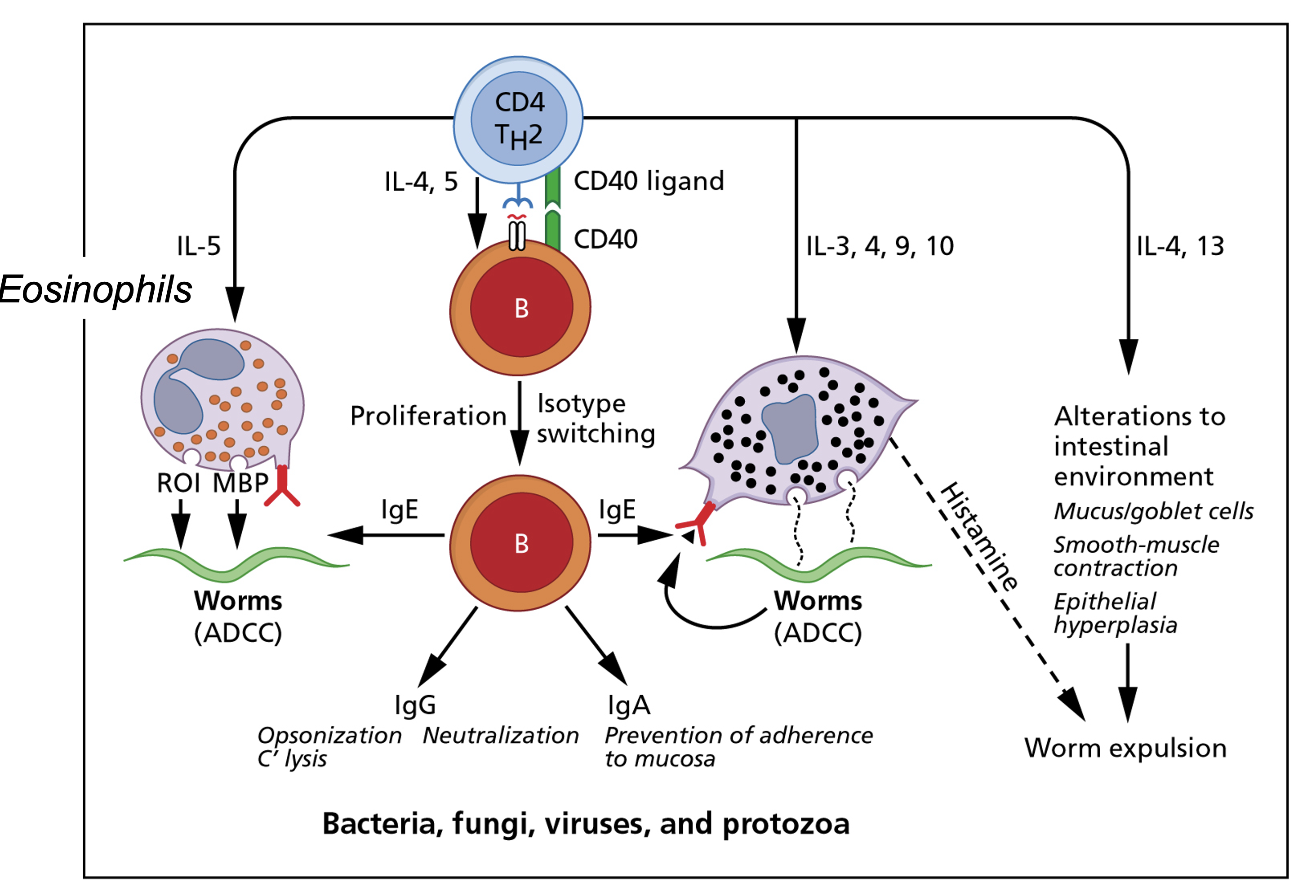
Th2 and CTL-driven immunity
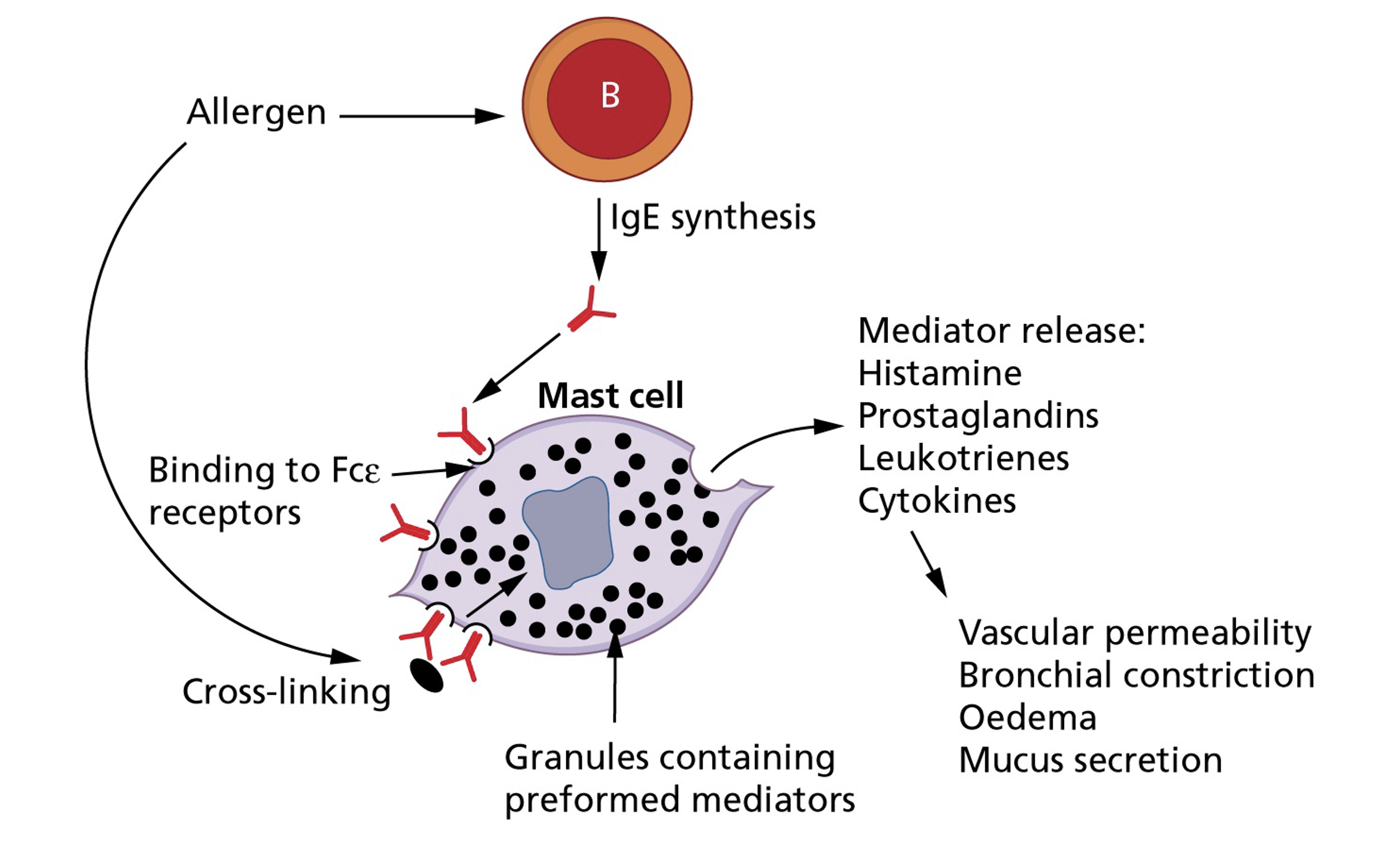
Cell-mediated driven allergy
Drugs that impair cell-mediated immunity
Common drugs that impair T-cell function:
- Corticosteroids
- Azathioprine, cyclosporine, tacrolimus
- mTOR inhibitors (sirolimus, everolimus)
- Purine analogues (fludarabine, cladribine)
- Alemtuzumab
Diseases: Hodgkin lymphoma, CLL
Examples of “Targeted therapy” risks
| Drug | Mechanism | Infection Risk |
|---|---|---|
| Ruxolitinib | JAK-STAT inhibitor | TB, HBV reactivation |
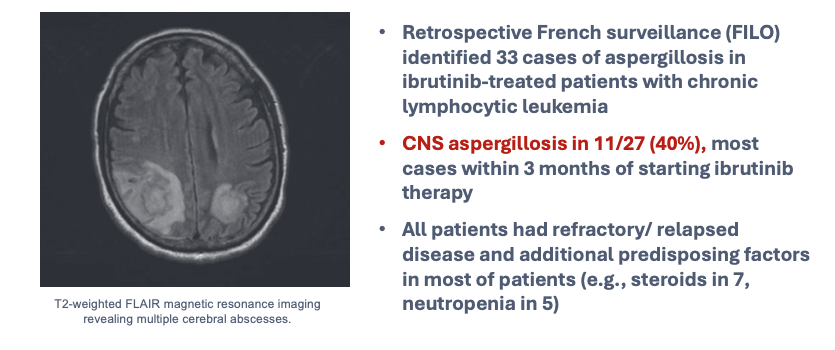
| Ibrutinib | BTK inhibitor | Aspergillosis, PJP |
| Idelalisib | PI3K inhibitor | P. jirovecii |
If you see a drug ending in “mab” or “nib” or “sib” ….consider unique infection risk
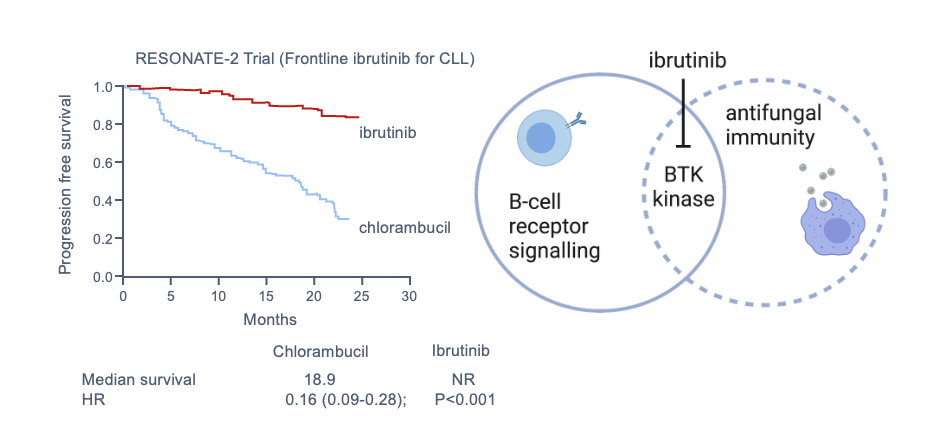
Ibrutinib
Unexpected fungal infections
Infectious Complications & Mortality
Humoral immunity
- B cells → antibody-secreting plasma cells
- Impaired in CLL, multiple myeloma
- Rituximab, CAR-T → B-cell depletion
- Profound, long-lasting hypogammaglobulinemia
Splenectomy: Loss of encapsulated bacteria defense-“Big 3”
Streptococcus pneumoniae
Haemophilus influenzae type B
Neisseria meningitidis
Less common: Capnocytophaga canimorsus, Salmonella spp. E. coli
PSV and PPSV23 vaccine, MENACWY and MenB vaccine, HIB, Influenzae- Patients should receive vaccines 2 weeks before elective splenectomy or 2 weeks after emergency splenetocmy
Physical Barriers
The Integument
Alimentary Tract
Immunodeficiency-Pathogen Associations
Neutropenia: Gram-positive pathogens
- Coagulase-negative staphylococci more common than
Staphylococcus aureus (most are from central venous catheter) - Viridans streptococci
- Enterococci
Neutropenia: Gram-negative pathogens
- Escherichia coli
- Pseudomonas aeruginosa
- Klebsiella pneumoniae
- Enterobacter spp.
Impaired cellular immunity
Impaired cellular immunity (viruses)
Impaired humoral immunity
Prevention Strategies
Pre-immunosuppression screening
Screen before starting immunosuppressive therapy:
| Pathogen | Test | Action if Positive |
|---|---|---|
| M. tuberculosis | IGRA (QuantiFERON) or TST | Isoniazid or rifampin prophylaxis |
| Hepatitis B | HBsAg, anti-HBc, anti-HBs | Antiviral prophylaxis (entecavir/tenofovir) |
| Hepatitis C | Anti-HCV, HCV RNA | Treat prior to immunosuppression if possible |
| HIV | 4th-gen Ag/Ab | ART optimization |
| Strongyloides | Serology (endemic areas/travel) | Ivermectin × 2 doses |
| T. cruzi | Serology (Latin American origin) | Benznidazole prophylaxis |
| VZV | IgG serology | Vaccinate if seronegative (pre-therapy) |
| Coccidioides/Histoplasma | Serology (endemic exposure) | Antifungal prophylaxis |
Prophylaxis principles
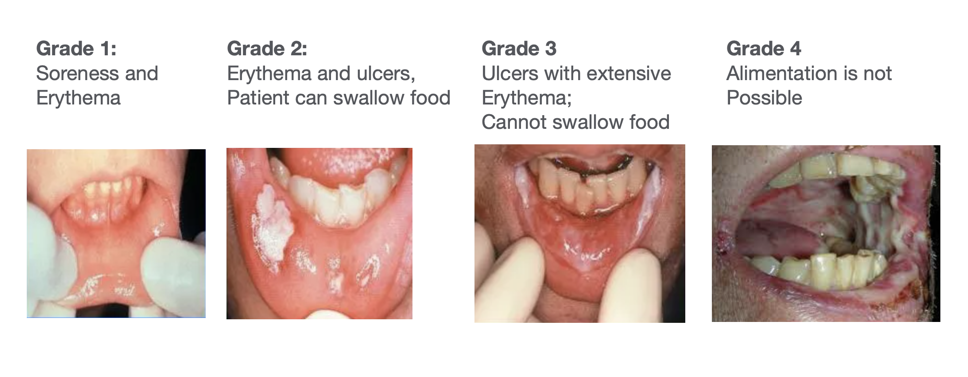
TMP-SMX for PJP :typically 1 DS tablet daily also covers:
- Toxoplasma
- S. aureus
- Nocardia
- Many gram-positives/negatives
Antiviral prophylaxis: Val(acyclovir) for CMV (weak activity), HSV, VZV prevention. Valganciclovir or letermovir for higher risk CMV patients
Antifungal prophylaxis
Risk-stratified approach:
High-risk indications (mold-active prophylaxis):
- AML induction/consolidation chemotherapy
- Allogeneic HSCT recipients
- Active GVHD on high-dose steroids
- Lung transplant recipients
Agents of choice: - Posaconazole (oral suspension or tablet) - Voriconazole (alternative) - Micafungin (IV, if oral not tolerated)
Lower-risk indications (Candida prophylaxis only):
- Autologous HSCT
- Prolonged neutropenia (>7 days)
- Mucositis + broad-spectrum antibiotics
Agents of choice: - Fluconazole - Micafungin
HBV reactivation prevention
A critical and underrecognized risk:
- HBV reactivation can occur with any immunosuppressive regimen
- Risk highest with: rituximab, anti-CD20 agents, anthracycline chemotherapy, corticosteroids ≥ 20 mg/day for ≥4 weeks, stem cell transplant
Who to treat:
- HBsAg(+): always give prophylaxis
- HBsAg(-)/anti-HBc(+): prophylaxis if high-risk regimen
- Reactivation mortality: up to 25% without prophylaxis
Prophylaxis options:
- Entecavir (preferred — high barrier to resistance)
- Tenofovir (TDF or TAF)
- Duration: continue 6–12 months after cessation of immunosuppression (18–24 months for anti-CD20)
Preemptive vs. prophylactic strategies for CMV
Two complementary approaches:
Prophylaxis (preferred when risk is high):
- Universal antiviral drug for defined at-risk period
- Letermovir (CMV D+/R− or R+ allo-HSCT)
- Valganciclovir (SOT, especially lung/heart recipients)
- Advantage: prevents CMV disease directly
- Disadvantage: late-onset CMV, drug toxicity, cost
Preemptive therapy (intermediate risk):
- Regular CMV PCR surveillance (every 1–2 weeks)
- Treat when viral load crosses threshold before disease
- Advantage: less antiviral exposure
- Disadvantage: requires rigorous monitoring; may miss rapid progressors
Vaccination: Solid Organ Transplant recipients
Core principle: Vaccinate before transplant whenever possible — responses are significantly blunted post-transplant on maintenance immunosuppression.
| Vaccine | Pre-transplant | Post-transplant | Notes |
|---|---|---|---|
| Influenza (inactivated) | ✅ Annually | ✅ Annually (≥1 month post-Tx) | Live attenuated influenza: contraindicated |
| Pneumococcal (PCV20) | ✅ ≥2 weeks before | ✅ ≥3–6 months post-Tx | Booster PPSV23 at 5 years if PCV15 series used |
| COVID-19 (mRNA) | ✅ | ✅ ≥1 month post-Tx; extra doses often needed | Check serology — responses frequently inadequate |
| Hepatitis B (double-dose) | ✅ Check anti-HBs | ✅ If not immune; confirm with anti-HBs | Anti-HBs target ≥10 IU/L |
| Tdap / Td | ✅ | ✅ ≥6 months post-Tx | Pertussis booster every 10 years |
| MMR, VZV (live) | ✅ ≥4 weeks before | ❌ Contraindicated post-transplant | Vaccinate seronegative candidates pre-listing |
| HPV | ✅ (if age-eligible) | ✅ ≥6 months post-Tx | 3-dose series; may respond poorly |
Vaccination: Hematopoietic stem cell transplant recipients
Core principle: HSCT ablates immune memory — recipients must be fully revaccinated post-transplant regardless of pre-transplant history.
| Vaccine | Timing Post-HSCT | Notes |
|---|---|---|
| Influenza (inactivated) | ≥6 months (or ≥4 months during seasonal outbreak) | Annual; live influenza contraindicated |
| Pneumococcal (PCV20/PCV15 × 3, then PPSV23) | Start at 3–6 months | 3-dose PCV series then PPSV23 ≥8 weeks later |
| COVID-19 (mRNA) | ≥3–6 months; 3-dose primary series | Check serology; additional doses often needed in GVHD |
| Hepatitis B (double-dose × 3) | ≥6 months | Confirm anti-HBs ≥10 IU/L; re-dose if inadequate |
| Tdap, then Td boosters | ≥6 months | 3-dose diphtheria/tetanus/pertussis series |
| Inactivated polio (IPV) | ≥6 months | 3-dose series; OPV contraindicated |
| Haemophilus influenzae b (Hib) | ≥6 months | 3-dose series; covers encapsulated bacteria |
| Meningococcal (ACWY + B) | ≥6 months | Important in asplenic/hyposplenic post-HSCT |
| MMR, VZV (live) | ≥24 months post-HSCT | Only if: off all immunosuppression, no active GVHD, CD4 ≥200 |
Immunoglobulin replacement therapy
Indications:
- Severe hypogammaglobulinemia (IgG <400 mg/dL) with recurrent infections
- Post-rituximab or CAR-T B-cell aplasia
- Common variable immunodeficiency (CVID)
- Post-HSCT (selected patients)
Products:
- IVIG (intravenous): 0.4–0.6 g/kg every 3–4 weeks
- SCIG (subcutaneous): home infusion, more stable IgG trough levels
Monitoring:
- Target trough IgG >500–700 mg/dL
- Adjust dose based on infection breakthrough
- Duration: until immune reconstitution (may be 1–3 years)
Environmental & infection control measures
Hospital setting (high-risk patients):
- HEPA-filtered rooms with positive pressure for prolonged neutropenia and allo-HSCT
- Avoid construction/renovation zones (aerosolized Aspergillus)
- Strict hand hygiene — single most effective infection prevention measure
- Central line bundles to reduce CLABSI
Dietary precautions (neutropenic diet — evidence limited but commonly practiced):
- Avoid raw/undercooked meat, eggs, unpasteurized products
- Wash fruits and vegetables thoroughly
- No well-ripened soft cheeses (Listeria)
Patient education
High-risk exposures to avoid:
- Gardening without protection (molds, Nocardia)
- Poor dental hygiene (Actinomyces, bacteremia)
- Marijuana smoking (Aspergillus)
- Raw seafood (Vibrio)
- Warm ocean swimming
Prophylaxis by immunodeficiency type
| Defect | Representative Conditions | Antibacterial | Antifungal | Antiviral | Other |
|---|---|---|---|---|---|
| Neutropenia (profound, >7d) |
AML induction, allo-HSCT engraftment | Levofloxacin† | Posaconazole (high-risk) / fluconazole | Acyclovir (HSV) | G-CSF if prolonged |
| T-cell deficiency | Allo-HSCT, calcineurin inhibitors, alemtuzumab, CAR-T | TMP-SMX (PJP, Nocardia, Toxoplasma) | Posaconazole / voriconazole | Letermovir or valganciclovir (CMV); acyclovir (HSV/VZV) | Screen for LTBI, Strongyloides; CMV surveillance |
| Humoral / B-cell deficiency | Rituximab, CAR-T, CLL, myeloma | TMP-SMX (PJP); consider azithromycin | Not routinely required | Acyclovir; monitor for enterovirus | IVIG if IgG <400 mg/dL + recurrent infections |
| Hypogammaglobulinemia | CVID, post-HSCT (late phase) | TMP-SMX or azithromycin | Not routinely required | Acyclovir | IVIG/SCIG replacement; PCV20, HIB, MenACWY vaccines |
| Asplenia / functional hyposplenism | Splenectomy, sickle cell, splenic irradiation | Penicillin V / amoxicillin (lifelong in many) | Not routinely required | Not routinely required | PCV20 + PPSV23, MenACWY + MenB, HIB, annual influenza; emergency antibiotic supply |
| Combined severe defect | Allo-HSCT + active GVHD, prolonged combination IS | TMP-SMX | Posaconazole (mold-active) | Letermovir / valganciclovir + acyclovir | IVIG; preemptive CMV PCR monitoring; TB/HBV prophylaxis as indicated |
Key Takeaways
Summary points
- 6% of population is immunocompromised
- Net state of immunosuppression = composite assessment
- First 100 days after transplant = highest risk period
- No single marker predicts infection risk
- Know pathogen associations with specific defects
- Prophylaxis significantly alters risk profile
Clinical Pearls
Remember
- TMP-SMX provides broader coverage than just PJP prophylaxis
- Timing matters—early vs late infections differ
- Targeted therapies (-mabs -nibs) have unexpected infection risks
- Consider the whole patient, not just the lab values

%20T%20Cell%20Therapy_%20Vein-to-Vein%20Process.png)