Penicillins and β-Lactamase Inhibitors
2026-04-14
Penicillins and β-Lactamase Inhibitors
Russell E. Lewis
Associate Professor of Infectious Diseases
Department of Molecular Medicine
University of Padua


russelledward.lewis@unipd.it
https://github.com/Russlewisbo
Slides and course materials: www.padovaid.com
Learning Objectives
By the end of this lecture, you should be able to:
- Describe the basic chemical structure of penicillins
- Explain how the side chain determines spectrum and stability
- Describe the mechanism of action involving PBPs
- Explain peptidoglycan synthesis and cross-linking
- List and explain the four mechanisms of β-lactam resistance
- Classify β-lactamases by Ambler molecular class
- Differentiate the five classes of penicillins by spectrum
- Describe pharmacokinetic properties and dosing adjustments
- Recognize adverse effects and hypersensitivity reactions
- Select appropriate β-lactam/β-lactamase inhibitor combinations
PART 1: History and chemistry
The Discovery of penicillin
From discovery to clinical use
| Year | Milestone |
|---|---|
| 1928 | Fleming discovers penicillin |
| 1929 | Fleming publishes findings |
| 1940 | Florey & Chain purify penicillin |
| 1941 | First human treated |
| 1943 | Mass production begins |
| 1945 | Nobel Prize awarded |
The Impact of penicillin
- Before penicillin: Minor infections could be fatal
- Bacterial endocarditis: 100% mortality
- Pneumococcal pneumonia: 30% mortality
- Staphylococcal sepsis: Often fatal
- Penicillin transformed infectious disease medicine
- Initiated the “antibiotic era”
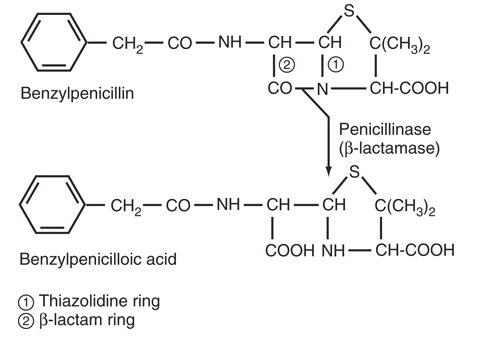
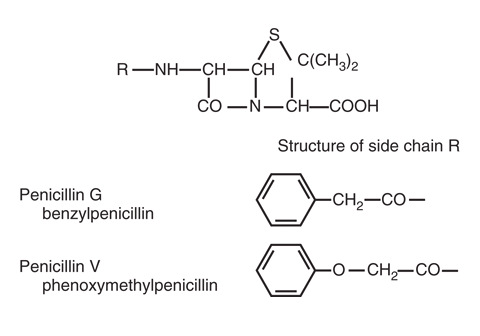
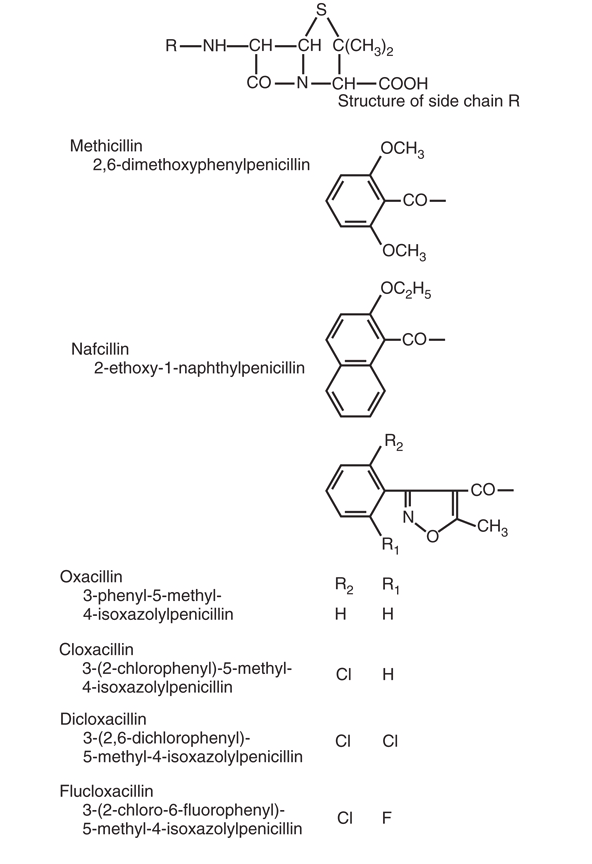
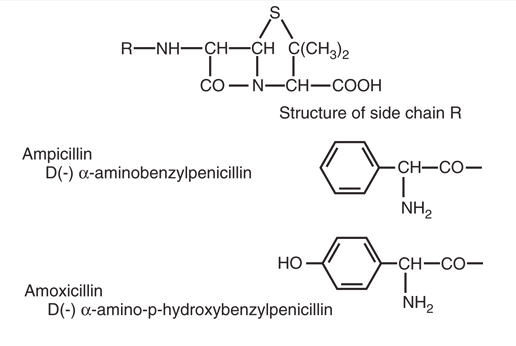
Basic penicillin structure
The three key structural components
- Thiazolidine ring
- 5-membered ring containing sulfur
- Provides structural stability
- β-Lactam ring
- 4-membered ring under strain
- ESSENTIAL for antibacterial activity
- Target of β-lactamases
- Side chain (R group)
- Variable component
- Determines properties of each penicillin
The β-Lactam ring: Why it matters
- Strained 4-membered ring — chemically reactive
- Mimics D-Ala-D-Ala terminus of peptidoglycan
- Covalently binds to PBP active site serine
- Opening the ring = loss of activity
- This is why β-lactamases cause resistance
Important
No β-lactam ring = No antibacterial activity
Classification of β-lactamases
| Ambler Class | Major Subtype | Preferred Substrates | Inhibitor | Genetic Localization | Representative Enzymes |
|---|---|---|---|---|---|
| A | Gram-positive β-lactamase 2a | Penicillins | Clavulanic acid | Chromosome or plasmid | PC1 |
| A | Gram-negative β-lactamase 2b | Penicillins, 1st-gen cephalosporins | Clavulanic acid | Plasmid or chromosomal | TEM-1, SHV-1 |
| A | Extended-spectrum β-lactamase 2be | Penicillins, extended-spectrum cephalosporins, aztreonam | Clavulanic acid | Plasmid | TEM-24, SHV-12, CTX-M-15 |
| A | Inhibitor-resistant TEM β-lactamase 2br | Penicillins | Clavulanic acidc | Plasmid | TEM-30, SHV-10 |
| A | Carbenicillin-hydrolyzing β-lactamase 2c | Carbenicillin | Clavulanic acidc | Plasmid | PSE-1, CARB-3 |
| A | Cephalosporin-hydrolyzing β-lactamase 2e | Extended-spectrum cephalosporins | Clavulanic acid | Chromosome | CepA |
| A | Carbapenem-hydrolyzing β-lactamase 2f | Carbapenems | Avibactam, Relebactam, Vaborbactam | Chromosome or plasmid | KPC-2, SME-1 |
| B | Metallo-β-lactamase 3a | All β-lactams except monobactam | EDTA, divalent cation chelators | Chromosome or plasmid | IMP-1, VIM-2, NDM-1 |
| C | AmpC-type β-lactamase | Cephalosporins | Cloxacillin, avibactam, relebactam, vaborbactam | Chromosome or plasmid | AmpC, CMY-2 |
The side chain: Determining properties
The side chain modifications determine:
- Acid stability — Can it survive gastric acid? (oral absorption)
- β-Lactamase stability — Is it protected from enzymatic destruction?
- Spectrum of activity — Which bacteria are susceptible?
- Protein binding — How much free drug is available?
- Tissue penetration — Where does the drug distribute?
- Cross-reactivity - Risk of allergies?
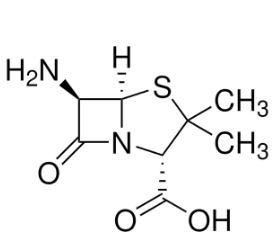
6-Aminopenicillanic Acid (6-APA)
PART 2: Mechanism of Action
Overview: How penicillins work
- Target: Final step of peptidoglycan synthesis
- Mechanism: Penicillins inhibit bacterial cell wall synthesis by binding to penicillin-binding proteins (PBPs)
- Result: Weakened cell wall
- Outcome: Osmotic lysis and cell death
- Effect: Bactericidal (kills bacteria)
Why bacteria need cell walls
- Bacteria have high internal osmotic pressure
- Without cell wall → osmotic lysis
- Cell wall = peptidoglycan polymer
- Gram-positive: thick layer (50-100 molecules)
- Gram-negative: thin layer (1-2 molecules) + outer membrane
- Human cells have no cell wall → selective toxicity
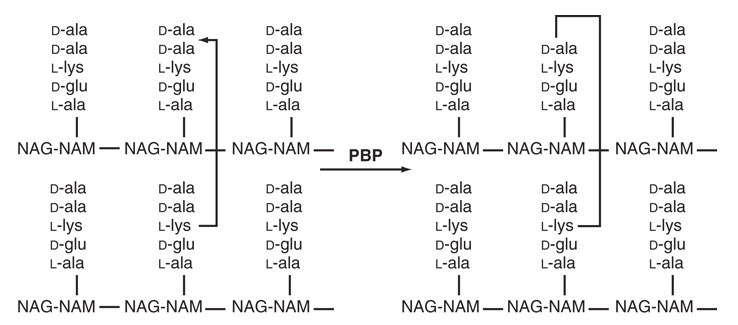
Peptidoglycan structure
- Backbone: Alternating NAG-NAM disaccharides
- NAG = N-acetylglucosamine
- NAM = N-acetylmuramic acid
- Pentapeptide stems: Attached to NAM
- Terminate in D-Ala-D-Ala
- Cross-links: Connect adjacent chains
- Provide strength and rigidity
Transpeptidation: The target reaction
- Pentapeptide ends in D-Ala-D-Ala
- Transpeptidase (PBP) binds to D-Ala-D-Ala
- Forms covalent intermediate with penultimate D-Ala
- Terminal D-Ala is released
- Cross-link formed with adjacent chain
- Penicillin mimics D-Ala-D-Ala and gets stuck
What are penicillin-binding proteins (PBPs)?
- Membrane-bound enzymes in all bacteria
- Catalyze final steps of cell wall synthesis
- Serine proteases — related to β-lactamases!
- Named for their ability to bind penicillin
- Multiple PBPs in each bacterial species
- Different PBPs have different functions
Classes of PBPs
| Class | Size | Function |
|---|---|---|
| High-MW Class A | >50 kDa | Bifunctional: transglycosylase + transpeptidase |
| High-MW Class B | >50 kDa | Transpeptidase only |
| Low-MW | <50 kDa | Carboxypeptidases |
PBP functions in E. coli
| PBP | Function | Inhibition result |
|---|---|---|
| PBP1a/1b | Transglycosylase + transpeptidase | Rapid cell lysis |
| PBP2 | Cell elongation, rod shape | Round cells (cocci) |
| PBP3 | Septum formation, cell division | Long filaments |
| PBP4-6 | Carboxypeptidases | Minor effects |
PBPs vs β-Lactamases: Key difference
PBPs (Cell Wall Synthesis)
- Bind penicillin tightly
- Slow deacylation rate
- Enzyme stays inhibited
- = Antibacterial effect
β-Lactamases (Resistance)
- Bind penicillin
- Fast deacylation rate
- Enzyme regenerates quickly
- = Drug destruction
Tolerance vs Resistance
- Resistance: Bacteria grow in presence of antibiotic
- MIC is high
- Tolerance: Bacteria survive but don’t grow
- MIC is low (susceptible)
- MBC is high (not killed)
- Examples: stationary phase cells, persisters in biofilm
Tip
Tolerance explains why some infections relapse despite “susceptible” organisms
PART 3: Resistance Mechanisms
Four mechanisms of β-Lactam resistance
- β-Lactamase production — enzymatic destruction
- Decreased permeability — porin mutations
- Efflux pumps — active drug removal
- Altered PBPs — low-affinity binding
Warning
Multiple mechanisms often coexist, especially in MDR gram-negatives
Mechanism 1: β-Lactamases
- Most common resistance mechanism
- Enzymes that hydrolyze the β-lactam ring
- Open the ring → inactive compound
- Gram-positive: Secreted extracellularly
- Gram-negative: Located in periplasmic space
- Can be chromosomal or plasmid-encoded
β-Lactamase mechanism
- β-Lactamase binds penicillin (like a PBP)
- Forms acyl-enzyme intermediate
- Rapid hydrolysis — water attacks the bond
- Ring opens, releasing penicilloic acid
- Enzyme regenerates immediately
- Cycle repeats (catalytic)
Ambler classification of β-Lactamases
| Class | Active Site | Mechanism | Examples |
|---|---|---|---|
| A | Serine | Acyl intermediate | TEM, SHV, CTX-M, KPC |
| B | Zinc (metallo) | Direct hydrolysis | NDM, VIM, IMP |
| C | Serine | Acyl intermediate | AmpC, CMY |
| D | Serine | Acyl intermediate | OXA enzymes |
Class A β-Lactamases: The most common
- TEM-1: Most common plasmid enzyme worldwide
- SHV-1: Common in Klebsiella
- CTX-M: Dominant ESBL globally
- KPC: Carbapenemase (major threat)
- Generally inhibited by clavulanic acid
- Exception: KPC (not well inhibited)
Extended-Spectrum β-Lactamases (ESBLs)
- Definition: Class A enzymes that hydrolyze extended-spectrum cephalosporins and aztreonam
- Evolution: Point mutations in TEM/SHV expanded spectrum
- CTX-M family: Now most common ESBL
- Inhibited by clavulanic acid (in vitro)
- Plasmid-encoded — spread easily
Warning
Clinical outcomes with BLI combinations may be unpredictable for serious ESBL infections
Class B: Metallo-β-Lactamases (MBLs)
- Use zinc ions instead of serine
- Hydrolyze all β-lactams EXCEPT aztreonam
- NOT inhibited by current approved inhibitors
- Examples: NDM-1, VIM, IMP
- Major global health threat
- Limited treatment options
Important
The “aztreonam loophole” — MBLs cannot hydrolyze monobactams
Class C: AmpC β-Lactamases
Cephalosporinases — preferentially hydrolyze cephalosporins
Often chromosomally encoded
Can be inducible (expressed when exposed to β-lactams)
Classic “SPACE” organisms: Serratia, Pseudomonas, Acinetobacter, Citrobacter, Enterobacter
Revised “HECK-YES” organisms: Hafnia, Enterobacter, Citrobacter (freundii complex), Klebsiella (aerogenes), Yersinia, Enterobacter, Serratia
NOT inhibited by clavulanic acid
Inhibited by avibactam, relebactam, vaborbactam
Class D: OXA enzymes
- Named for ability to hydrolyze oxacillin
- Heterogeneous group with variable spectra
- Some are carbapenemases (OXA-48, OXA-23)
- OXA-48: Poorly inhibited by most inhibitors
- Exception: Avibactam inhibits OXA-48
- Increasingly important in Acinetobacter (OXA-23)
Carbapenemases: The greatest threat
| Enzyme | Class | Distribution | Inhibitors |
|---|---|---|---|
| KPC | A | Americas, worldwide | Avibactam, vaborbactam, relebactam |
| NDM | B | South Asia, global | None currently approved |
| VIM | B | Europe, global | None currently pproved |
| OXA-48 | D | Middle East, Europe | Avibactam |
Mechanism 2: Decreased permeability
Mechanism 3: Efflux Pumps
Mechanism 4: Altered PBPs
- PBPs with low affinity for β-lactams
- Drug binds poorly → ineffective inhibition
- Examples:
- MRSA: Acquires PBP2a (mecA gene)
- Penicillin-resistant pneumococci: Mosaic PBP genes
- Enterococcus faecium: Low-affinity PBP5
Important
MRSA resistance is NOT due to β-lactamases — it’s due to PBP2a
MRSA: Mechanism of resistance
- MRSA carries mecA gene (on SCCmec element)
- mecA encodes PBP2a (also called PBP2’)
- PBP2a has very low affinity for β-lactams
- Native PBPs are inhibited, but PBP2a continues working
- Cell wall synthesis continues
- All β-lactams ineffective (except ceftaroline, ceftobiprole)
PART 4: Classification of Penicillins
Five classes of penicillins
- Natural penicillins — Penicillin G, Penicillin V
- Penicillinase-resistant — Nafcillin, Oxacillin, Dicloxacillin
- Aminopenicillins — Ampicillin, Amoxicillin
- Carboxypenicillins — Ticarcillin (obsolete)
- Ureidopenicillins — Piperacillin
Class 1: Natural penicillins
Natural Penicillins: Spectrum
Excellent activity:
- Streptococcus pyogenes (Group A strep)
- Streptococcus agalactiae (Group B strep)
- Streptococcus pneumoniae (susceptible strains)
- Neisseria meningitidis
- Treponema pallidum (syphilis)
- Most oral anaerobes
- Listeria monocytogenes
Natural Penicillins: Clinical uses
| Infection | Drug of Choice |
|---|---|
| Group A strep pharyngitis | Penicillin V |
| Syphilis (all stages) | Penicillin G |
| Neurosyphilis | IV Penicillin G |
| Meningococcal meningitis | Penicillin G |
| Actinomycosis | Penicillin G |
| Gas gangrene (C. perfringens) | Penicillin G |
Class 2: Penicillinase-resistant penicillins
Nafcillin, Oxacillin, Dicloxacillin, Flucloxacillin
- Bulky side chains create steric hindrance
- Resist hydrolysis by staphylococcal β-lactamases
- Spectrum: MSSA and streptococci
- No gram-negative activity
- NOT effective against MRSA
Antistaphylococcal penicillins: Details
| Drug | Route | Protein Binding | Elimination | Special Considerations |
|---|---|---|---|---|
| Nafcillin | IV | 90% | Hepatic | Hypokalemia, phlebitis |
| Oxacillin | IV | 90% | Mixed | Hepatotoxicity |
| Dicloxacillin | PO | 96% | Renal/hepatic | Highest protein binding |
| Flucloxacillin | PO/IV | 96% | Renal | Not available in US |
Class 3: Aminopenicillins
Aminopenicillins: Expanded spectrum
- Same as natural penicillins PLUS:
- Enterococcus faecalis
- Haemophilus influenzae (non-β-lactamase producing)
- Escherichia coli (non-β-lactamase producing)
- Proteus mirabilis
- Salmonella and Shigella spp.
- Listeria monocytogenes
Ampicillin vs amoxicillin
| Property | Ampicillin | Amoxicillin |
|---|---|---|
| Oral absorption | 30-55% | 74-92% |
| Effect of food | Decreased | None |
| Preferred route | IV | Oral |
| Bioequivalence | — | Better than ampicillin |
Tip
For oral therapy, amoxicillin is almost always preferred
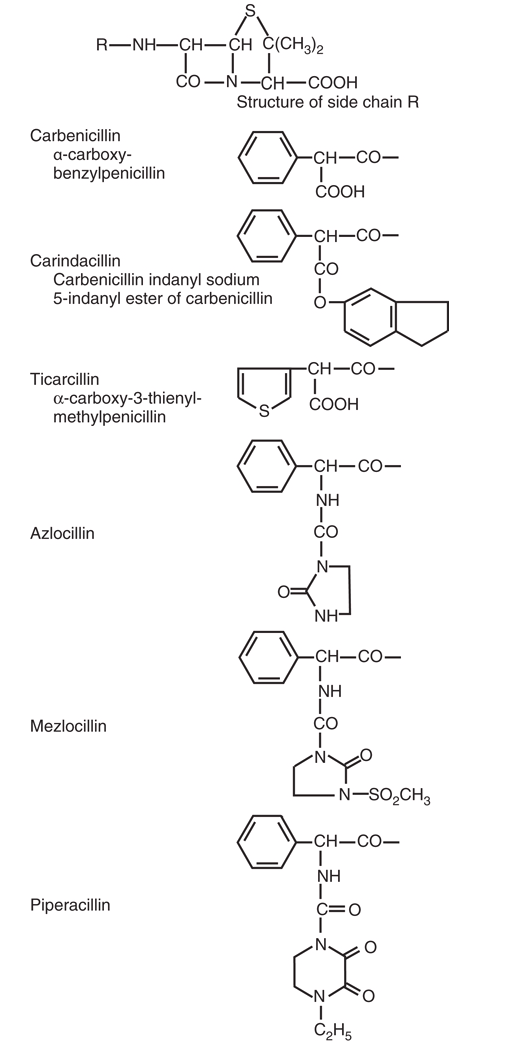
Classes 4 & 5: Antipseudomonal penicillins
Spectrum summary table
| Class | Gram+ | Gram- | Pseudomonas | Anaerobes | MRSA |
|---|---|---|---|---|---|
| Natural | +++ | - | - | ++ | - |
| Antistaphylococcal | ++ (Staph) | - | - | - | - |
| Aminopenicillins | ++ | + | - | ++ | - |
| Antipseudomonal | + | ++ | ++ | +++ | - |
PART 5: Pharmacokinetics
Oral absorption
| Penicillin | Absorption (%) | Food Effect |
|---|---|---|
| Penicillin V | 60 | None |
| Ampicillin | 30-55 | Decreased |
| Amoxicillin | 74-92 | None |
| Dicloxacillin | 37 | Decreased |
| Flucloxacillin | 44 | Decreased |
:::{style=“width: fit-content; margin: auto;”} ::: {.callout-tip} Amoxicillin has the best oral bioavailability ::: :::
Protein binding
- Ranges from 17% (aminopenicillins) to 97% (dicloxacillin)
- Only free drug is active
- High protein binding:
- Reduces active drug concentration
- Prolongs half-life
- May reduce tissue penetration
- Clinical significance debated
Distribution
- Generally good tissue penetration
- Achieve therapeutic levels in:
- Lung, liver, kidney, muscle
- Pleural, peritoneal, synovial fluid
- Bone (variable)
- Poor penetration without inflammation:
- CNS, eye, prostate
CNS Penetration
| Condition | Penicillin G | Ampicillin |
|---|---|---|
| Normal meninges | <1% | <1% |
| Inflamed meninges | 5-10% | 13-14% |
Note
Inflammation is required for adequate CNS levels. As meningitis resolves, drug penetration decreases.
Elimination
- Most penicillins: Primarily renal excretion
- Glomerular filtration + tubular secretion
- Short half-lives (0.5-1.5 hours)
- Probenecid can block tubular secretion
- Exceptions:
- Nafcillin: Primarily hepatic
- Oxacillin: Mixed hepatic/renal
Renal dosing: When to adjust
| CrCl (mL/min) | Adjustment Needed? |
|---|---|
| >50 | Usually no |
| 30-50 | Consider for some agents |
| 10-30 | Yes for most agents |
| <10 | Yes, significant reduction |
| Hemodialysis | Dose after dialysis |
Dosing in renal failure
| Agent | CrCl 10-29 | Hemodialysis |
|---|---|---|
| Penicillin G | 75% dose | Dose post-HD |
| Ampicillin | 0.5-2g q12h | 0.5-1g q12-24h |
| Amoxicillin | 500mg q12h | 500mg q12-24h |
| Piperacillin | 3g q8-12h | 3g q12h |
| Nafcillin | No change | No change |
Optimizing β-Lactam dosing
- β-Lactams are time-dependent killers
- Efficacy correlates with %T>MIC (time above MIC)
- Target: 40-70% of dosing interval above MIC
- Strategies to optimize:
- More frequent dosing
- Extended infusions (3-4 hours)
- Continuous infusions
Extended infusion dosing
Example: Piperacillin-tazobactam
| Traditional | Extended Infusion |
|---|---|
| 4.5g over 30 min q6h | 4.5g over 4 hours q8h |
| Higher peak, lower trough | Lower peak, higher trough |
| Less time above MIC | More time above MIC |
Tip
Extended infusion may improve outcomes, especially for organisms with higher MICs
PART 6: Adverse effects
Overview of adverse effects
- Hypersensitivity reactions — most important
- Gastrointestinal effects
- Hematologic effects
- Neurologic effects
- Nephrotoxicity
- Hepatotoxicity
- Electrolyte disturbances
Hypersensitivity: Types
| Type | Timing | Mechanism | Manifestations |
|---|---|---|---|
| Type I | Minutes-hours | IgE-mediated | Anaphylaxis, urticaria, angioedema |
| Type II | Days | Antibody-mediated | Hemolytic anemia, cytopenia |
| Type III | 1-3 weeks | Immune complex | Serum sickness, drug fever |
| Type IV | Days-weeks | T-cell mediated | Maculopapular rash, contact dermatitis |
Penicillin allergy: By the numbers
- ~10% of patients report penicillin allergy
- <1% have true IgE-mediated allergy when tested
- ~2% will react if challenged
- True anaphylaxis: <0.01%
- Allergy often wanes over time
- 50% lose sensitivity within 5 years
- 80% lose sensitivity within 10 years
Penicillin allergy de-labeling
- History assessment — Was it really an allergic reaction?
- Risk stratification — High risk vs low risk features
- Skin testing — Detects IgE-mediated allergy
- Graded oral challenge — Confirms tolerance
- Update medical record — Remove incorrect allergy label
Tip
De-labeling programs are safe and improve patient care
Cross-reactivity with cephalosporins
- Historical estimates: 10% cross-reactivity (overestimate)
- Current data: ~1-2% cross-reactivity
- Cross-reactivity relates to side chain similarity
- Highest risk: Similar R1 side chains
- Ampicillin → Cephalexin, Cefadroxil
- Lower risk: Dissimilar side chains
- Ceftriaxone, cefepime
Other adverse effects
| Effect | Most Common With | Notes |
|---|---|---|
| Diarrhea | Ampicillin, amoxicillin-clav | Disruption of gut flora |
| C. difficile | All | Risk with any antibiotic |
| Neutropenia | Prolonged high-dose therapy | Reversible |
| Seizures | High-dose penicillin G | Especially in renal failure |
| Interstitial nephritis | Methicillin, nafcillin | Allergic mechanism |
Agent-specific toxicities
- Nafcillin: Hypokalemia, phlebitis
- Oxacillin: Hepatotoxicity, interstitial nephritis
- Ampicillin: Maculopapular rash (especially with EBV)
- Amoxicillin-clavulanate: Diarrhea, hepatotoxicity
- Piperacillin: Platelet dysfunction, hypokalemia
- High-dose Penicillin G (K+ salt): Hyperkalemia
PART 7: β-Lactamase inhibitors
β-Lactamase inhibitor classes
Traditional (β-lactam)
- Clavulanic acid
- Sulbactam
- Tazobactam
Novel (non-β-lactam)
- Avibactam
- Relebactam
- Vaborbactam
Mechanism: Traditional inhibitors
- Inhibitor binds to β-lactamase active site
- Forms stable acyl-enzyme complex
- Complex undergoes irreversible fragmentation
- Enzyme is permanently inactivated
- “Suicide inhibitor” mechanism
- One inhibitor molecule = one enzyme molecule
Traditional inhibitor combinations
| Inhibitor | Partner | Formulations |
|---|---|---|
| Clavulanic acid | Amoxicillin | Oral, IV |
| Sulbactam | Ampicillin | IV |
| Tazobactam | Piperacillin | IV |
Spectrum of traditional inhibitors
Inhibited:
- Class A β-lactamases (TEM, SHV, many ESBLs)
- S. aureus β-lactamase
- Bacteroides β-lactamase
NOT inhibited:
- Class B (metallo-β-lactamases)
- Class C (AmpC)
- KPC (weak inhibition)
Novel β-Lactamase inhibitors
| Inhibitor | Partner | Unique Feature |
|---|---|---|
| Avibactam | Ceftazidime | Inhibits KPC, OXA-48, AmpC |
| Relebactam | Imipenem | Inhibits KPC, AmpC |
| Vaborbactam | Meropenem | Inhibits KPC, AmpC |
Important
None inhibit metallo-β-lactamases (NDM, VIM, IMP)
Inhibitor spectrum summary
| Inhibitor | Class A | ESBLs | KPC | AmpC | MBLs | OXA-48 |
|---|---|---|---|---|---|---|
| Clavulanate | ✓ | ± | ✗ | ✗ | ✗ | ✗ |
| Tazobactam | ✓ | ± | ✗ | ✗ | ✗ | ✗ |
| Avibactam | ✓ | ✓ | ✓ | ✓ | ✗ | ✓ |
| Vaborbactam | ✓ | ✓ | ✓ | ✓ | ✗ | ✗ |
MIC values: BLI combinations
| Organism | Amp/Amox | Amox-Clav | Pip-Tazo |
|---|---|---|---|
| S. aureus (MSSA) | 16 | 1 | 1 |
| H. influenzae (BL+) | >16 | 0.5 | 0.06 |
| E. coli | >16 | 4 | 2 |
| K. pneumoniae | >16 | 2 | 4 |
| B. fragilis | >16 | 0.5 | 2 |
| P. aeruginosa | >16 | >16 | 4 |
Clinical limitations of BLI combinations
Warning
In vitro activity does not guarantee clinical success
- ESBL infections: Treatment failures reported with BLI combinations
- Inoculum effect: High bacterial loads may overwhelm inhibitor
- Serious infections: Carbapenems often preferred for ESBL bacteremia
- AmpC producers: Traditional inhibitors ineffective
PART 8: Clinical Application
Clinical Case 1: Skin Infection
55-year-old man with cellulitis
- Erythema, warmth, swelling of left lower leg
- No drainage, no crepitus
- No systemic symptoms
- No diabetes, no recent hospitalization
- No history of MRSA
What is the most appropriate oral therapy?
Case 1: Answer
- Likely pathogens: S. aureus, Group A strep
- MRSA risk: Low (no risk factors)
- Best choice: Dicloxacillin 500mg QID or Cephalexin 500mg QID
- Rationale: Narrow spectrum, covers MSSA + strep
- Avoid: Amoxicillin-clavulanate (too broad), Fluoroquinolones (no added benefit)
Clinical Case 2: Pneumonia
68-year-old woman with community-acquired pneumonia
- Cough, fever, dyspnea for 3 days
- CXR: Right lower lobe infiltrate
- O2 sat 94% on room air
- COPD (mild), no recent antibiotics
- Outpatient treatment appropriate
What is the most appropriate oral therapy?
Case 2: Answer
- Most likely pathogen: S. pneumoniae
- Other possibilities: H. influenzae, atypicals
- COPD: Increases H. influenzae risk
- Best choice: Amoxicillin-clavulanate 875mg BID + Azithromycin (for atypicals)
- Alternative: Respiratory fluoroquinolone (moxifloxacin)
- Rationale: Covers pneumococcus (including most resistant strains), H. influenzae
Clinical Case 3: UTI
32-year-old woman with uncomplicated cystitis
- Dysuria, frequency for 2 days
- No fever, no flank pain
- No recent antibiotics
- No structural urinary abnormalities
What is the most appropriate therapy?
Case 3: Answer
- Most likely pathogen: E. coli (75-95% of uncomplicated UTIs)
- Resistance concern: >40% of E. coli resistant to ampicillin
- First-line options (per guidelines):
- Nitrofurantoin 100mg BID x 5 days
- TMP-SMX (if local resistance <20%)
- Fosfomycin single dose
- Amoxicillin/ampicillin: NOT first-line due to resistance
Clinical Case 4: Intra-Abdominal Infection
52-year-old man with perforated appendicitis
- Post-operative day 1, now febrile
- WBC 18,000
- CT shows pelvic abscess
- No recent antibiotics, no recent hospitalization
What is the most appropriate empiric therapy?
Case 4: Answer
- Pathogens: Gram-negatives (Enterobacterales), Anaerobes (B. fragilis), possibly Enterococcus
- Best choice: Piperacillin-tazobactam 4.5g IV q6h (extended infusion)
- Alternative: Ceftriaxone + metronidazole
- Rationale: Broad gram-negative coverage + anaerobes
- Duration: Until source controlled, then course completion
Clinical Case 5: Endocarditis
45-year-old man with S. aureus bacteremia
- IV drug user, new murmur
- TEE: Tricuspid vegetation
- Blood culture: MSSA (oxacillin MIC 0.25)
- No contraindications to β-lactams
What is the most appropriate therapy?
Case 5: Answer
- Diagnosis: MSSA tricuspid valve endocarditis
- Duration: 4-6 weeks IV therapy
- Best choice:* Cefazolin 2 g IVq8h less nephrotoxicity (Burdet et al. 2025)and possibly better outcomes (Prosty et al. 2025; McDanel et al. 2017)than Nafcillin 2g IV q4h or Cloxacillin 2g IV q4h
- NOT vancomycin: Inferior outcomes for MSSA
- Monitoring: Renal function, signs of drug fever, eosinophilia
Important
For MSSA, nafcillin/oxacillin are superior to vancomycin
Clinical Case 6: Meningitis
22-year-old college student with meningitis
- Headache, fever, stiff neck, rash
- CSF: 2000 WBC (95% PMN), protein 250, glucose 20
- Gram stain: Gram-negative diplococci
- Suspected Neisseria meningitidis
What is the most appropriate therapy?
Case 6: Answer
- Diagnosis: Meningococcal meningitis
- Best choice: CCeftriazone 2 grams IV q12h
- Alternative: Penicillin G 4 million units IV q4h
- Duration: 7 days
- Chemoprophylaxis: Close contacts need rifampin, ciprofloxacin, or ceftriaxone
- Dexamethasone: Controversial for meningococcus (consider for pneumococcus)
Clinical case 7: Syphilis
28-year-old man with primary syphilis
- Painless chancre on penis
- RPR reactive, TP-PA positive
- Reports penicillin allergy: “rash as a child”
What is the most appropriate management?
Case 7: Answer
- Treatment of choice: Benzathine penicillin G 2.4 million units IM x 1
- Alternative Ceftriaxone 2 grams IV daily 10-14 days
- Penicillin allergy: Likely not true allergy (childhood rash)
- Management options:
- Penicillin skin testing → if negative, treat with penicillin
- Penicillin desensitization → then treat
- NOT acceptable: Doxycycline or azithromycin (inferior efficacy)
Quick selection guide
| Infection | First-Line Penicillin |
|---|---|
| Strep pharyngitis | Penicillin V |
| MSSA skin/soft tissue | Dicloxacillin |
| MSSA bacteremia/endocarditis | Nafcillin |
| CAP (outpatient, no comorbidity) | Amoxicillin |
| CAP (outpatient, with COPD) | Amoxicillin-clavulanate |
| Intra-abdominal infection | Piperacillin-tazobactam |
| Syphilis | Benzathine penicillin G |
| Listeria meningitis | Ampicillin |
Summary and Key Takeaways
Key point 1: Structure determines function
- β-Lactam ring is essential for activity
- Side chain determines spectrum, stability, and pharmacokinetics
- Modifications create different penicillin classes
- Understanding structure explains clinical properties
Key point 2: Know the resistance mechanisms
- β-Lactamases: Most common; hydrolize β-lactam ring
- Permeability: Porin changes in gram-negatives
- Efflux: Active drug removal
- Altered PBPs: Low-affinity binding (MRSA, PRP)
- Multiple mechanisms often coexist
Key point 3: Match spectrum to pathogen
- Natural penicillins: Streptococci, syphilis, meningococcus
- Antistaphylococcal: MSSA only
- Aminopenicillins: Add enterococci, some gram-negatives
- Pip-tazo: Broad including Pseudomonas, anaerobes
- None work against MRSA
Key point 4: β-Lactamase inhibitors have limits
- Traditional inhibitors: Class A only
- NOT effective against:
- AmpC (Class C)
- Metallo-β-lactamases (Class B)
- KPC (limited)
- Newer inhibitors: Broader coverage but still gaps
- In vitro activity ≠ clinical success
Key point 5: De-Label penicillin allergies
- Most reported allergies are NOT true allergies
- True IgE-mediated allergy is rare (<1%)
- Skin testing can identify true allergy
- De-labeling improves patient care
- Don’t avoid penicillins unnecessarily