Fever of Unknown Origin
2026-03-01
Fever of Unknown Origin (FUO)
Prof. Russell E. Lewis
Department of Molecular Medicine
University of Padua
russelledward.lewis@unipd.it
https://github.com/Russlewisbo
slides available at: www.padovaid.com
 |
Objectives
- Recognize leading infectious causes of FUO in key patient groups
- Identify fever patterns and clinical histories that may direct diagnosis
- Differentiate FUO risks and possible spectrum of pathogens in immunocompromised hosts
The history of fever
- 10th Century BCE Persian Physician Akhawayni defined a system for fever curves in Hidāyat al-Muta’allimīn fī al-Tibb (The Student’s Handbook of Medicine)
- Hippocratic physicians proposed that body temperature, and physiologic harmony in general, involved a delicate balance among four corporal humors—blood, phlegm, black bile, and yellow bile.
- Fever was due to excess of yellow bile (many infections caused jaundice)
- Galen: many types of fever developed from putrefaction of humors.
- Middle ages: demonic possession
- 18th century (Harvey’s discovery of circulation)- friction of blood flow through body causing fermentation and putrefaction in intestines
- Claude Bernard in the 19th century- metabolic processes in the body
Febris - Roman Goddess of Fever
The legend of Febris was said to center around the haunting marshes of Camagna in Southern Italy where like clockwork every year, the people would become deathly ill with a mysterious disease. She was so feared by the Romans that the suffering population had created a cult to Febris. They went so far as to wear protective amulets and build her temples in order to worship her to win her favour.
Early thermometers
A typical design of a thermoscope is a tube in which a liquid rises and falls as the temperature changes. The Sanctorius thermoscope. Source: Professor Francis Ring, the University of Leeds
Fever in modern medicine
Wunderlich’s pioneering studies of thermometry reported normal temperature at 37°C
Since the 19th century, humans have become gradually colder - 0.05° to 0.5°C per decade!
- Current normal range is 36.3 to 36.5°C
- Less manual labor, less chronic inflammation-infection, poor dental health, increased body mass
Mackowiak (1992): mean oral temperature 36.8 ± 0.4°C; only 8% had 37°C
Fever now defined as: early-morning temperature ≥37.2°C or anytime ≥ 37.8°C
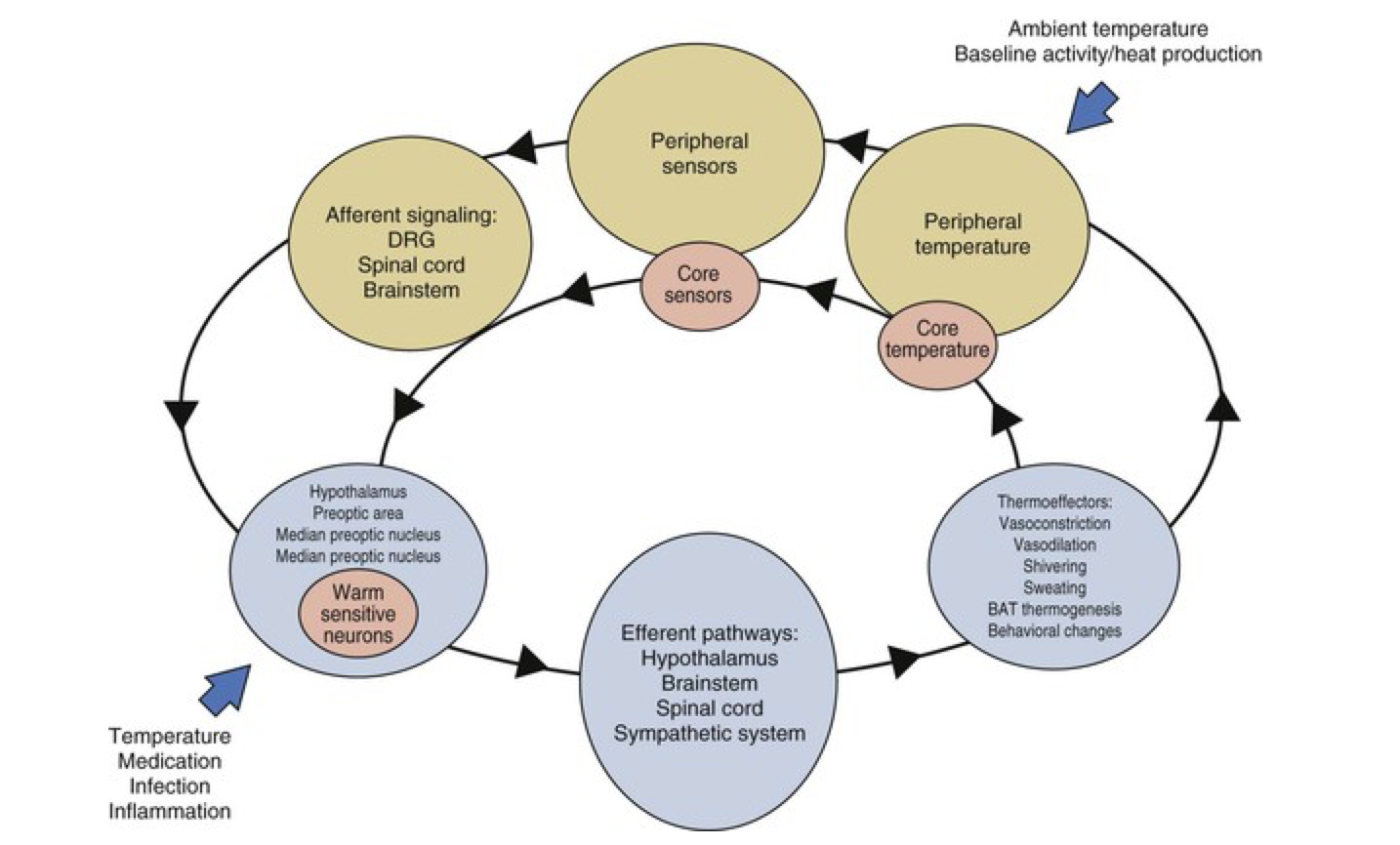
Thermal homeostasis
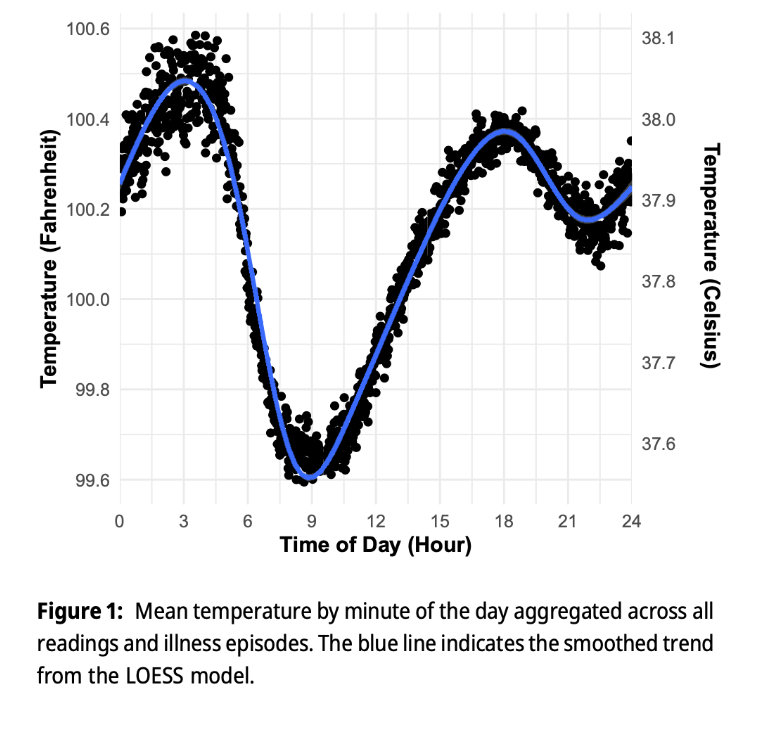
Diurinal pattern of body temperature
Physiologic variables affecting body temperature
- Age and sex differences
- Women ~0.5°C higher at ovulation
- Infants have higher baseline temperatures
- Circadian rhythm
- Zenith: late afternoon (peak)
- Nadir: early morning (trough)
- Other factors
- Exercise and physical exertion
- Medications (antipyretics, corticosteroids)
- Digestion and recent meals
- Chronic disease and metabolic conditions
Fever vs. Hyperthermia
| Feature | Fever | Hyperthermia |
|---|---|---|
| Set point | Elevated | Normal |
| Mechanism | Regulated response | Unregulated heat generation |
| Cause | Pyrogens (LPS, IL-1, TNF) | Heat exposure, drugs, malignant hyperthermia |
| Pathophysiology | Altered hypothalamic setpoint | Failure of heat dissipation |
| Sweating | Absent initially (shivering); later excessive |
May be absent |
| Treatment response | Antipyretics effective | Antipyretics ineffective; cooling required |
| Examples | Infection, NIID, malignancy | Heat stroke, neuroleptic malignant syndrome |
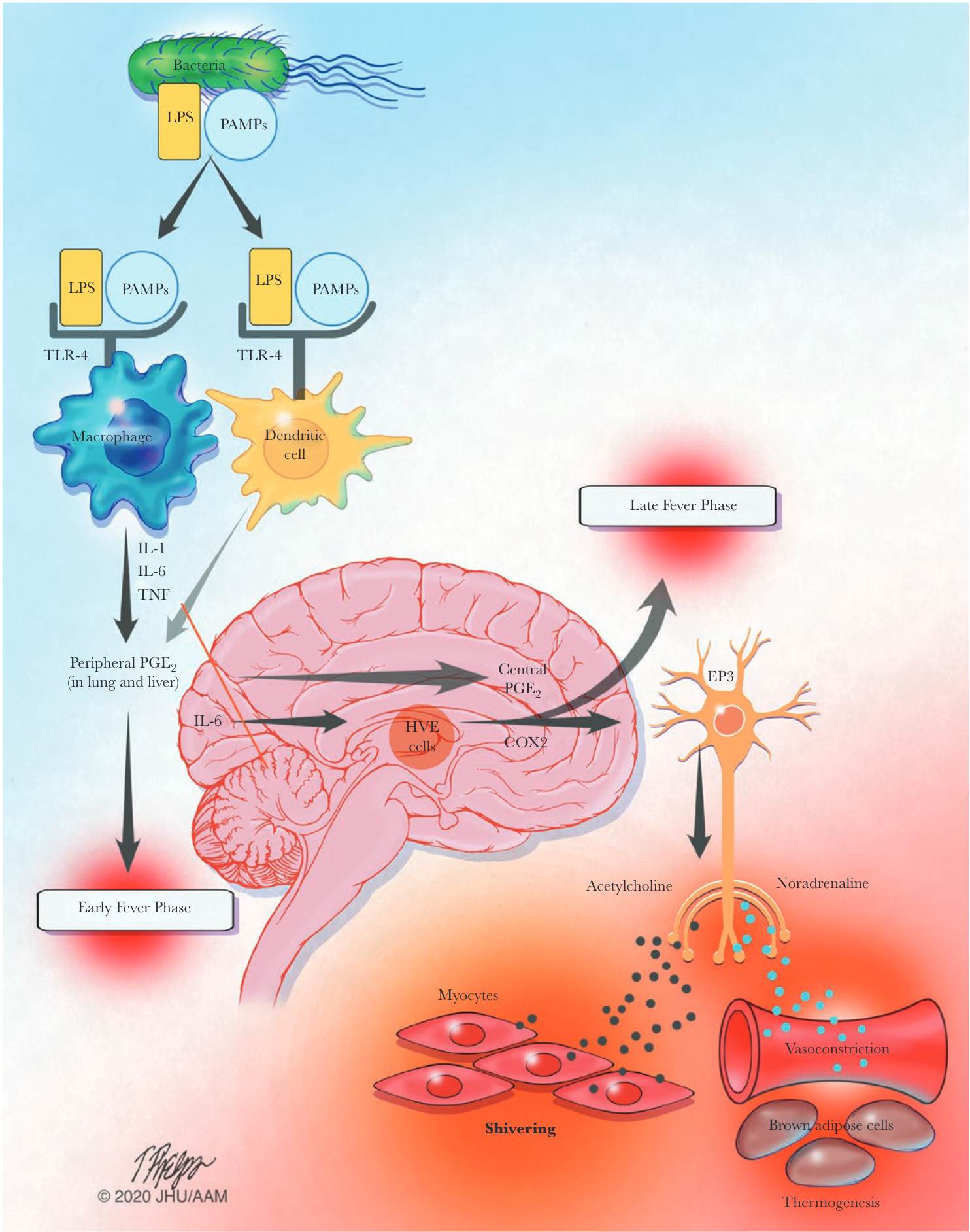
Infection-associated fever - The febrile response
Sequelae of fever - Benefits of fever
- Phylogenetic conservation suggests fever is beneficial - appears in ectotherms and endotherms
- Microbial metabolism - most pathogenic bacteria are mesophiles (optimal growth at 35-37°C)
- Even modest temperature elevations slow replication
- Iron sequestration - fever triggers hepatic synthesis of hepcidin and lactoferrin
- Sequesters free iron necessary for bacterial replication
- Enhanced immune function
- Increased neutrophil migration
- Augmented antibody production
- Enhanced T-cell proliferation
Acute phase proteins
| Category | Proteins |
|---|---|
| ↑ Complement system | C3, C4, C9, Factor B, C1 inhibitor, C4b-binding protein, Mannose-binding lectin |
| ↑ Coagulation / fibrinolysis | Fibrinogen, Plasminogen, tPA, Urokinase, Protein S, Vitronectin, PAI-1 |
| ↑ Antiproteases | α₁-Protease inhibitor, α₁-Antichymotrypsin, Pancreatic trypsin inhibitor, Inter-α-trypsin inhibitors |
| ↑ Transport proteins | Ceruloplasmin, Haptoglobin, Hemopexin |
| ↑ Inflammatory mediators | Secreted PLA₂, LPS-binding protein, IL-1 receptor antagonist, G-CSF |
| ↑ Others | CRP, Serum amyloid A, α₁-Acid glycoprotein, Fibronectin, Ferritin, Angiotensinogen |
| ↓ Negative acute-phase | Albumin, Transferrin, Transthyretin, α₂-HS glycoprotein, Alpha-fetoprotein, Thyroxine-binding globulin, IGF-I, Factor XII |
Acute phase phenomena
| Category | Phenomena |
|---|---|
| Neuroendocrine | Fever, somnolence, anorexia; ↑ CRH, corticotropin & cortisol; ↑ arginine vasopressin; ↓ IGF-I; ↑ adrenal catecholamines |
| Hematopoietic | Anemia of chronic disease; leukocytosis; thrombocytosis |
| Metabolic | Muscle loss & negative nitrogen balance; ↓ gluconeogenesis; osteoporosis; ↑ hepatic lipogenesis; ↑ adipose lipolysis; ↓ lipoprotein lipase activity; cachexia |
| Hepatic | ↑ Metallothionein, iNOS, heme oxygenase, MnSOD, TIMP-1; ↓ phosphoenolpyruvate carboxykinase activity |
| Nonprotein plasma | Hypozincemia, hypoferremia, hypercupremia; ↑ plasma retinol & glutathione |
Epidemiology of FUO - Definitions
| Category | Key causes / definition |
|---|---|
| Classic FUO | Infection (TB, endocarditis, occult abscess, zoonoses, enteric fever, syphilis, histoplasmosis), malignancy, autoimmune/autoinflammatory, miscellaneous; includes virally suppressed HIV (CD4 >200) |
| Nosocomial FUO | FUO arising in hospitalized patients |
| ICU | Bacteremia, pneumonia, C. difficile, fungemia, catheter infections, PE, acalculous cholecystitis, drug fever, stroke/intracranial hemorrhage |
| Non-ICU | Similar to ICU causes; patient not critically ill |
| Immunodeficiency-associated FUO | Highly variable; depends on type and degree of immunodeficiency |
| Organ-transplant recipients | Viruses, donor-derived infections, Strongyloides hyperinfection, opportunistic fungi, rejection, GVHD, HLH, ureaplasma hyperammonemia |
| Neutropenia | Febrile >5 days despite empirical antibiotics; influenced by neutropenia duration, GVHD prophylaxis, antimicrobial agents |
| HCT recipients | Pre-engraftment: neutropenic causes; early post-engraftment: herpesvirus, adenovirus, hyperacute GVHD, pneumonia; late: relapsed cancer, immune reconstitution |
| HIV/AIDS (no ART) | Acute retroviral syndrome, mycobacteria, endemic mycoses, toxoplasmosis, cryptococcosis, HHV-8–associated diseases, lymphoma |
| Travel-associated FUO | Malaria, enteric fever, leptospirosis, viral hemorrhagic fevers, typhus, undifferentiated tropical febrile illness |
Fever therapy to treat neurosyphilis
Treponema pallidum is uniquely sensitive to increased temperatures
Fever therapy involved purposely infecting the patient with Plasmodium vivax with control of the infection with anti- malarians while maintaining the fever it causes to the detriment of other, ongoing, and then-incurable infections such as late-stage syphilis
This type of pyrotherapy was most famously used by psychiatrist Julius Wagner-Jauregg, who won the Nobel Prize for Medicine in 1927 for his elaboration of the procedure in treating neurosyphilis
Classic FUO
- Definition:
- Temperature of > 38.3°C > 3 weeks
- Fever >2 separate outpatient visits with diagnostic investigations or
- Fever >2 visits in hospital of 3 days with diagnostic investigations
- However,… these definitions are largely subjective
- Leading causes:
- Infections (geography dependent)
- Inflammatory conditions (age dependent)
- Cancer (age dependent)
- Undiagnosed/unknown
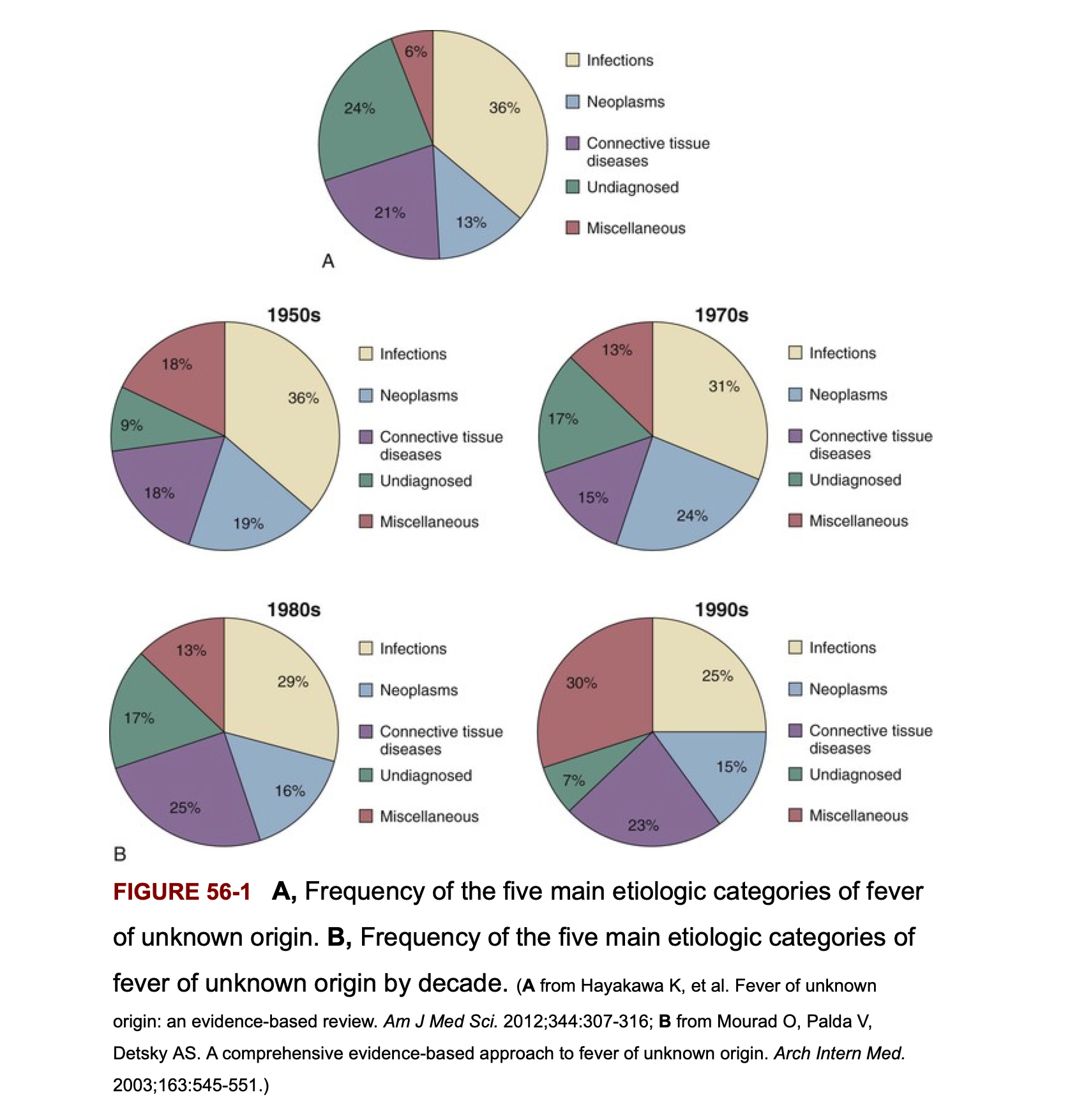
Frequency of the 5 main etiologic categories of FUO
Classic FUO - Infectious Etiology
- Chronic or relapsing infections
- Occult abscess (intra-abdominal, hepatic, splenic, renal, perirectal)
- Endocarditis (both native and prosthetic valve)
- Tuberculosis (pulmonary and extrapulmonary)
- Complicated urinary tract infections (pyelonephritis, prostatitis)
- Osteomyelitis (especially vertebral)
- Other: brucellosis, leptospirosis, syphilis, bartonellosis
Rare and miscellaneous causes of fever
| Category | Examples |
|---|---|
| Autoinflammatory / periodic fevers | Adult-onset Still’s disease, Behçet’s syndrome, Familial Mediterranean fever, Familial Hibernian fever, Periodic fever, Schnitzler’s syndrome |
| Vascular / cardiac | Aortic dissection, Aortitis, Atrial myxoma, Giant coronary aneurysm, Pericarditis, Postpericardiotomy syndrome, Polyarteritis nodosa, Pulmonary emboli, Veno-occlusive disease |
| Hematologic / oncologic | Castleman’s disease, Cyclic neutropenia, Hemoglobinopathies, Hemolytic anemias, Hemophagocytic syndrome, Histiocytosis X, Immunoblastic lymphadenopathy, Lymphomatoid granulomatosis, Myeloproliferative syndromes, Paroxysmal hemoglobinurias, Rosai-Dorfman disease, TTP |
| Granulomatous / lymphoproliferative | Allergic alveolitis, Granulomatous hepatitis, Granulomatous peritonitis, Kikuchi-Fujimoto disease, Lofgren syndrome, Retroperitoneal fibrosis, Sarcoidosis, Subacute necrotizing lymphadenitis |
| Autoimmune / rheumatologic | Autoimmune cholangitis, Erythema multiforme, Inflammatory bowel disease, Serum sickness, Sjögren’s syndrome, Wegener’s granulomatosis |
| Endocrine / metabolic | Addison’s disease, Fabry’s disease, Parathyroid apoplexy, Pheochromocytoma, Thyroiditis & thyrotoxicosis, Vitamin B₁₂ deficiency |
| GI / hepatic | Alcoholic hepatitis, Cirrhotic fever, Pancreatitis |
| Infectious | Bartonellosis, Carcinomatous meningitis, Chronic meningitis, Hantavirus, Human picornavirus, Hypereosinophilic syndrome, Infected urachal cyst, Sinusitis, Whipple’s disease |
| Other | Drug fever & hypersensitivities, Factitious fever, Metal fume fever, Resorbing hematoma |
Uncommon and rare causes of FUO
| Category | Examples |
|---|---|
| Infections | Bartonellosis, hantavirus, coccidioidomycosis, histoplasmosis, blastomycosis, cysticercosis |
| NIID | Addison disease, Behçet syndrome, SLE, vasculitis, inflammatory bowel disease, GPA, sarcoidosis, Sjögren syndrome, thyroiditis, adult-onset Still disease |
| Malignancy | Atrial myxoma, lymphoma, leukemia, pheochromocytoma, Schnitzler syndrome |
| Other | Cirrhotic fever, drug fever, factitious fever, vitamin B₁₂ deficiency, pulmonary embolism |
Classic FUO in infants and children
- Respiratory tract infections - viral, atypical organisms
- Other infections:
- UTIs (especially important in young girls)
- Brucellosis, tuberculosis, bartonellosis
- Systemic inflammatory conditions:
- Kawasaki disease (critical, age < 5 years)
- Inflammatory bowel diseases
- Still’s disease (juvenile rheumatoid arthritis)
- Note: Connective tissue diseases and cancers are generally rare in children
- Important: Joint involvement is a sign of serious disorder - consider endocarditis, leukemia, connective tissue disease
Classic FUO in elderly patients
- In developed countries: connective tissue diseases > infections
- Temporal arteritis (critical diagnosis - risk of blindness)
- Polymyalgia rheumatica
- Other vasculitides
- Diagnostic challenge: symptoms are subacute and non-specific, easily attributed to aging
- Infections still need to be considered:
- Intra-abdominal abscess
- Complicated UTIs (often without pyuria)
- Tuberculosis (may be reactivation)
- Endocarditis (prosthetic valves more common in elderly)
- Malignancy: solid tumors and hematologic malignancies increase with age
Fever in returning travelers
| Diagnosis | Maclean et al (n = 587) | Doherty et al (n = 195) |
|---|---|---|
| Malaria | 32% | 42% |
| Respiratory tract infection | 11% | 2.6% |
| Dysentery | 4.5% | 5.1% |
| Urinary tract infection / pyelonephritis | 4% | 2.6% |
| Dengue fever | 2% | 6.2% |
| Enteric fever | 2% | 1.5% |
| Hepatitis | 6% | 3% |
| Tuberculosis | 1% | 2% |
| Rickettsial infection | 1% | 0.5% |
| Amebic liver abscess | 1% | 0% |
| Acute HIV infection | 0.3% | 1% |
| Other miscellaneous infections | 4.3% | 9.2% |
| Miscellaneous noninfectious causes | 6% | 1% |
| Undiagnosed | 25% | 24.6% |
Nosocomial (Health-Care Associated) FUO
- Leading causes:
- Drug fever (especially to antibiotics, anti-epileptic medications)
- Post-operative complications (e.g., occult abscess, anastomotic leak)
- Decubitus ulcers with superimposed infection
- Septic thrombophlebitis (peripherally inserted central catheters)
- Recurrent pulmonary emboli
- Myocardial infarction
- Hematologic malignancy (newly diagnosed during hospitalization)
- Blood transfusion reaction
- Reactions to contrast media used in radiologic procedures
- Clostridium difficile colitis
Fever in post-operative patients
- Epidemiology: > 1/3 of patients manifest fever in first 5 days post-surgery
- Infectious vs. non-infectious: < 10% of febrile patients have an identified source or positive cultures
- Pathophysiology: Fever may represent a physiological response to surgically-induced tissue injury
- Release of pyrogenic cytokines and interleukins
- Not necessarily indicative of infection
- Clinical pearl: Early fever (post-op day 1-3) is usually non-infectious; later fever (day 5+) warrants infection investigation
FUO in ICU patients
- Early fevers are common and often non-infectious
- Associated with good prognosis
- Related to inflammatory response to critical illness
- Prolonged fever carries a poorer prognosis
- Common complications:
- Sinusitis (from mechanical ventilation, supine positioning, feeding tubes)
- Ventilator-associated pneumonia
- Catheter-related bloodstream infections
- Other causes similar to nosocomial FUO
- Abscess, drug fever, septic thrombophlebitis, pulmonary emboli
FUO in stroke patients
- Non-infective fevers are common in stroke patients
- Occur earlier after stroke than infection
- Related to hypothalamic injury and cytokine release
- Infection-related complications:
- UTI (from urinary catheterization)
- Aspiration pneumonia
- DVT/PE
- Clinical challenge: Distinguishing fever from infarct-related inflammation vs. infection can be difficult; broad-spectrum antibiotics are often given empirically
FUO in neutropenic patients
ANC = Total WBC x (% Segs + % Bands)
Definition of neutropenia:
- ANC < 500 cells/mm³
- “Profound” neutropenia: ANC < 100 cells/mm³
Frequency of fever during chemotherapy-induced neutropenia:
- 10-50% of patients with solid tumors
- 80% of those with hematologic malignancies during ≥1 chemotherapy cycle
- Most patients will have NO infectious etiology documented
Critical finding: Signs of inflammation are notoriously absent other than fever
- No exudate, no fluctuance, no purulent drainage
- Makes clinical diagnosis extremely difficult
Clinical manifestations of infection related to absolute neutrophil count
| Sign/Symptom | Type of Infection | % with ANC<100 | % with ANC>1000 |
|---|---|---|---|
| Fever | Overall | 98 | 76 |
| Bacteremia | Overall | 43 | 13 |
| Fluctuance | Anorectal | 8 | 67 |
| Exudate | Skin | 5 | 92 |
| Purulent sputum | Pneumonia | 8 | 84 |
| Pyuria | UTI | 11 | 97 |
Possible causes of fever in neutropenic patients not responding to broad-spectrum antibiotics
| Cause | Approx. frequency in high-risk patients |
|---|---|
| Fungal infections susceptible to empirical therapy | 40% |
| Bacterial infections (cryptic foci, biofilms, resistant organisms) | 10% |
| GVHD after hematopoietic stem cell transplantation | 10% |
| Fungal infections resistant to empirical antifungal therapy | 5% |
| Toxoplasma gondii, mycobacteria, or fastidious pathogens (Legionella, Mycoplasma, Chlamydia pneumoniae, Bartonella) | 5% |
| Viral infections (herpesviruses, CMV, EBV, HHV-6, VZV, HSV, parainfluenza, RSV, influenza) | 5% |
| Undefined (drug fever, chemotherapy toxicity, antitumor responses, undefined pathogens) | 25% |
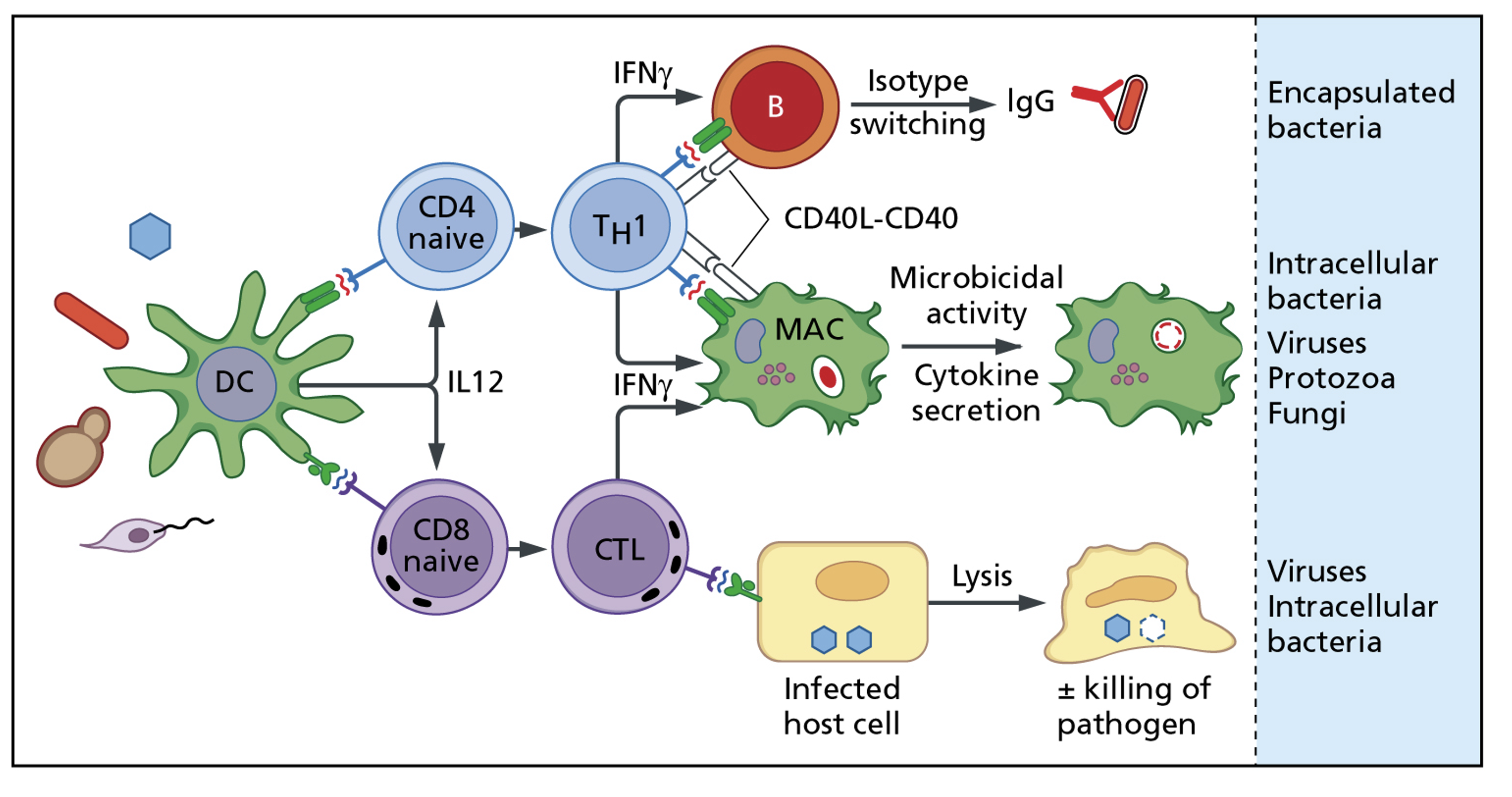
Cell-mediated immunity - Th1 pathway
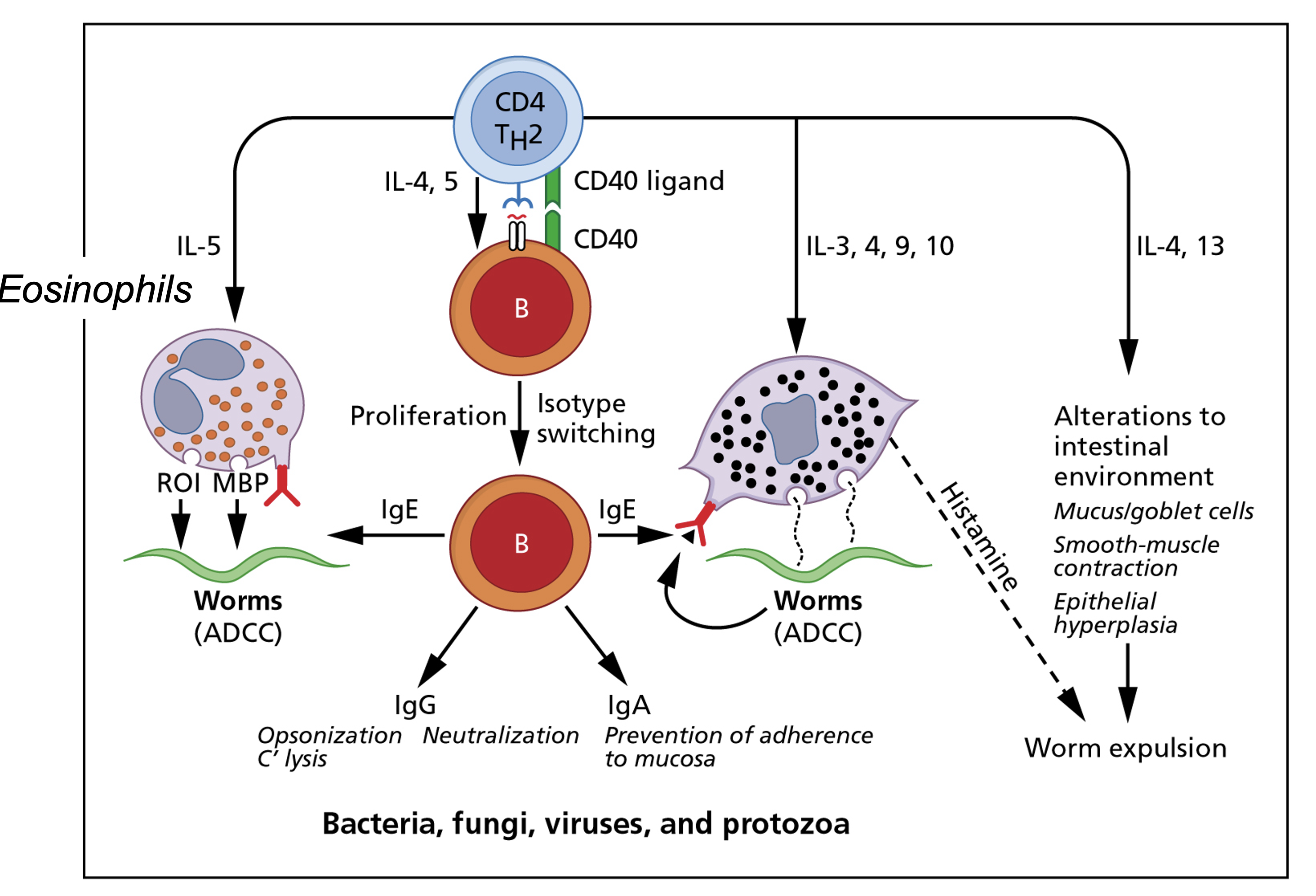
Cell-mediated immunity - Th2 pathway
Cell-mediated immunity -
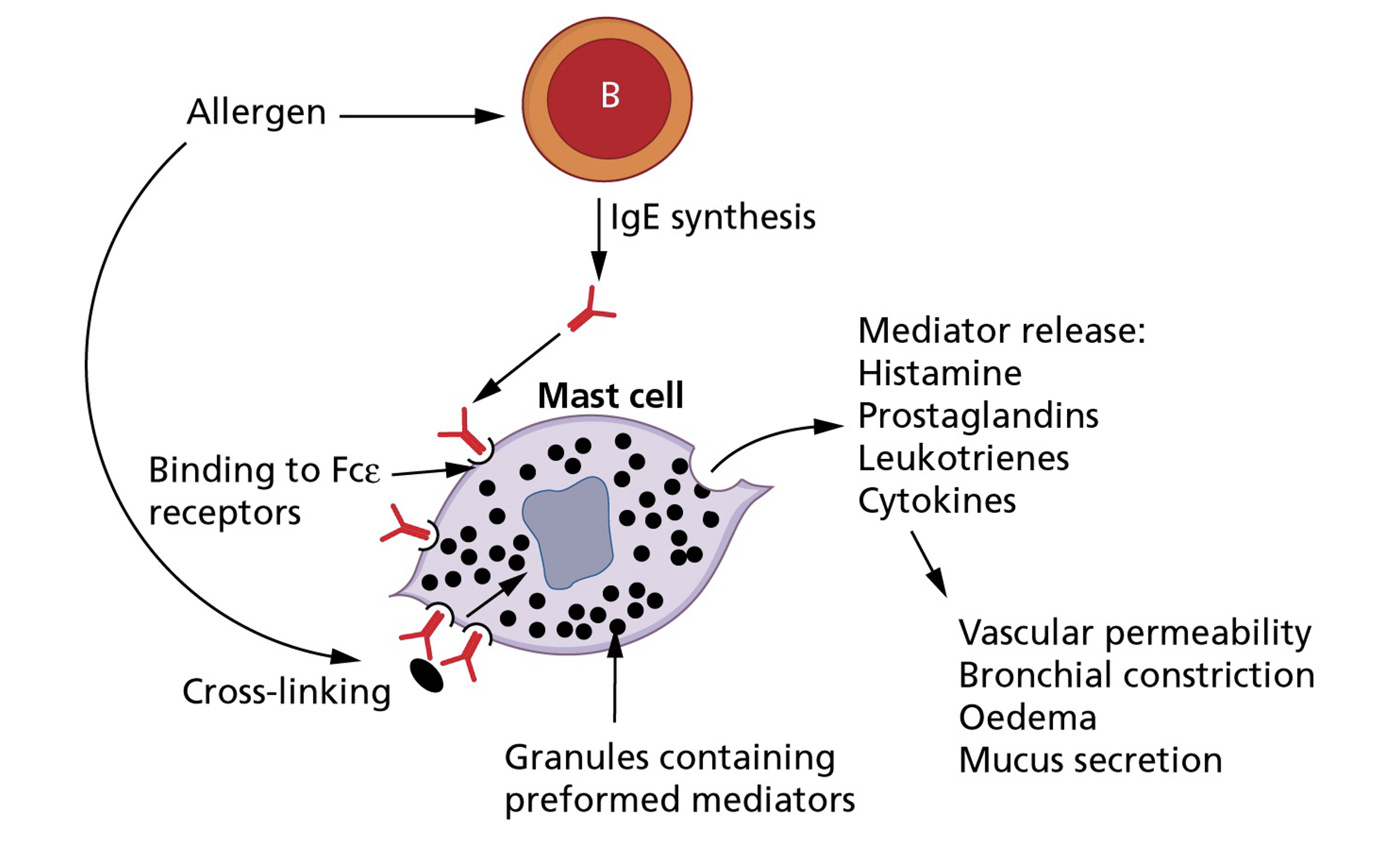
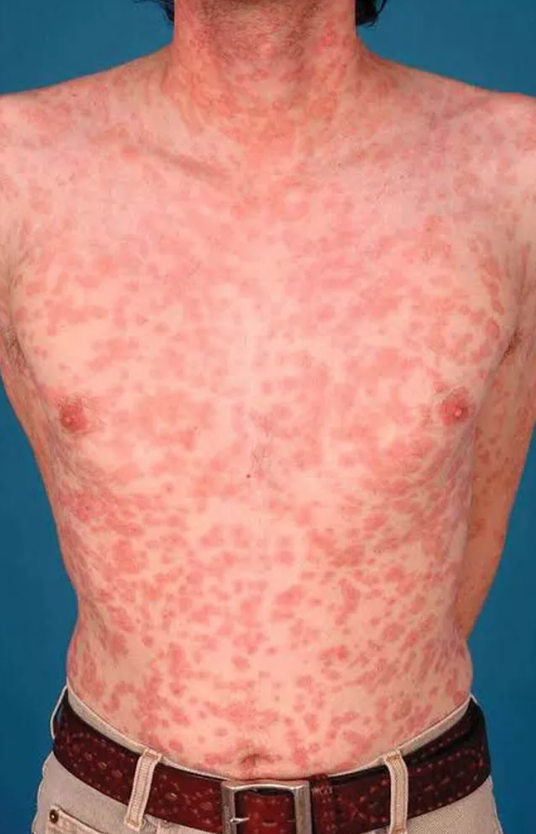
Drug allergy (Type IV hypersensitivity)
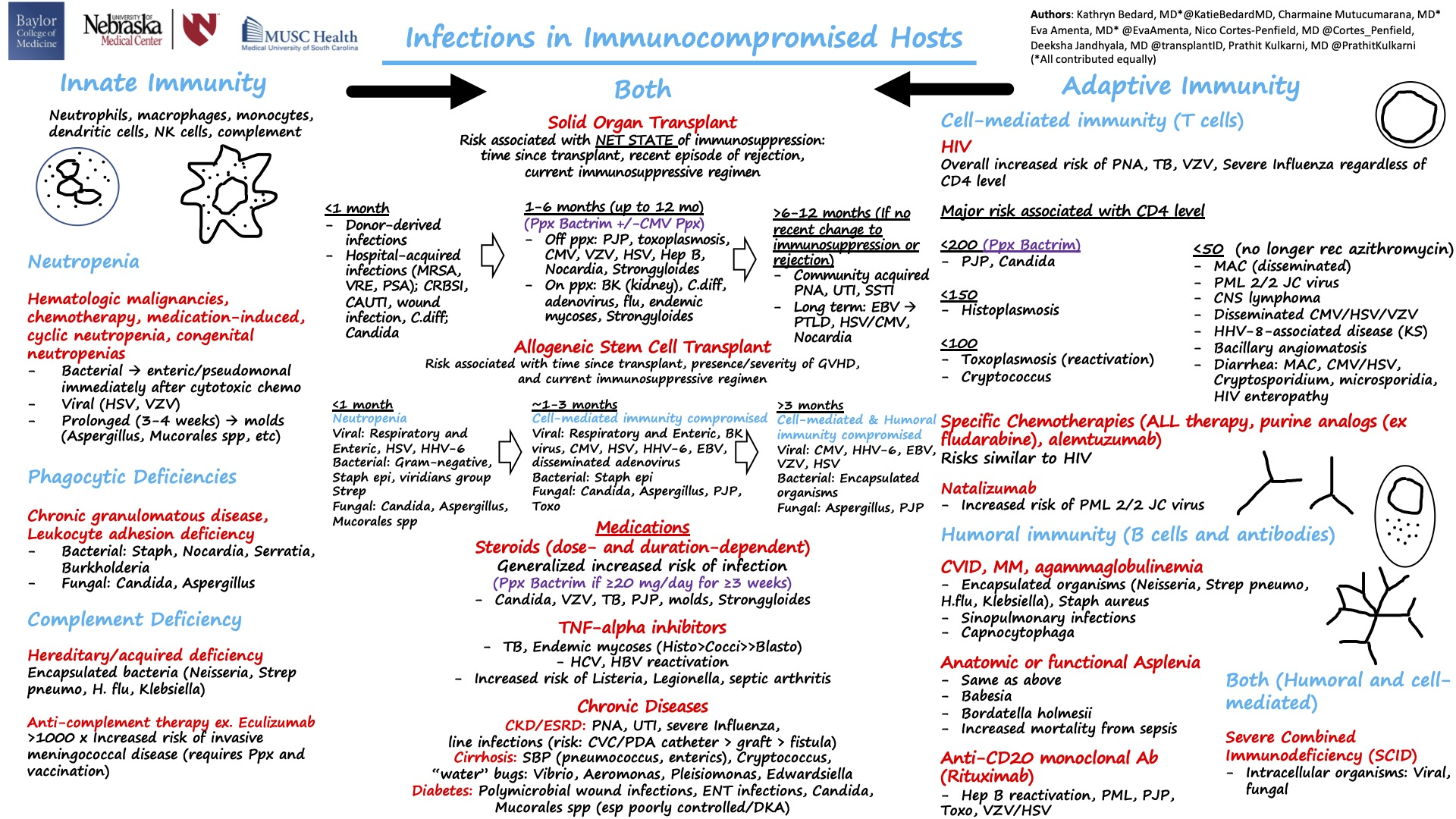
Infections in immunocompromised hosts:
A summary in one slide!
HIV-related FUO
- Primary infection phase characterized by mononucleosis-like illness
- Fever is common
- May be undiagnosed if it precedes seroconversion (acute retroviral syndrome)
- Later phases of untreated HIV
- Episodes of fever become common
- Often signify superimposed illness - e.g., opportunistic infections
- Infections may manifest atypically
- HAART era impact
- Once highly-active antiretroviral therapy (HAART) is started
- Effective suppression of HIV viral load
- Frequency of FUO falls markedly
- CD4
+recovery allows control of opportunistic infections
Etiology of fever in HIV-Associated FUO (n=70)
| Etiology | No. (%) |
|---|---|
| Infection | 63 (88%) |
| DMAC | 22 (31%) |
| Pneumocystis jirovecii pneumonia | 10 (13%) |
| CMV | 8 (11%) |
| Histoplasmosis | 5 (7%) |
| Viral (not CMV) | 5 (7%) |
| Bacterial | 4 (5%) |
| Mycobacterium tuberculosis | 4 (5%) |
| Fungal (not histoplasmosis) | 2 (3%) |
| Parasitic | 2 (3%) |
| Mycobacterium genavense | 1 (1%) |
| Neoplasia | 6 (8%) |
| Lymphoma | 5 (7%) |
| Kaposi sarcoma | 1 (1%) |
| Miscellaneous | 3 (4%) |
| Drug fever | 2 (3%) |
| Castleman disease | 1 (1%) |
Naproxen (NSAID) fever suppression test for “tumor fever”
- Indications for trial:
- Temperature > 37.8°C at least once daily for ≥2 weeks
- Lack of evidence of infection (physical exam, labs, imaging)
- Absence of drug fever, transfusion reaction, or allergic mechanisms
- Lack of response to ≥7 days of empiric antibiotics
- Procedure:
- Naproxen 500 mg BID × 3-5 days
- Positive result:
- Prompt complete defervescence (lysis of fever)
- Sustained normal temperature while receiving naproxen
- Fever recurrence when drug discontinued
- Note: Not universally used; sensitivity/specificity debated
Diagnosis of FUO
General diagnostic evaluation of FUO
| Step | Investigation |
|---|---|
| History & Exam | Comprehensive history; repeated physical exams |
| Laboratory | CBC, comprehensive metabolic panel, urinalysis with microscopy |
| Inflammatory Markers | ESR, C-reactive protein |
| Autoimmune Screening | ANA, rheumatoid factor |
| Imaging - First Line | Chest radiograph, CT abdomen/pelvis |
| Cultures | Blood cultures (3 specimens without antimicrobials), urine culture |
| Serologies | CMV IgM/PCR, heterophil antibody (EBV) in young adults |
| Tuberculosis | Tuberculin skin test, interferon-gamma release assay |
| Advanced Imaging | MRI, PET-CT, duplex ultrasound lower extremities |
| Invasive Procedures | Biopsy (lymph node, liver, bone marrow) if indicated |
Patient history
Helps guide choice of initial laboratory investigations - this is the most important step
Travel history: where, when, duration, exposures to animals, arthropod vectors, contaminated water
Exposure to animals and work environment: pet birds (psittacosis), cats (toxoplasmosis, bartonellosis), tick exposure (Lyme, Q fever)
Recent contact with ill persons or family history of FUO (e.g., familial Mediterranean fever)
Complete medication list: including OTC drugs, supplements, recent antimicrobials
Prior history of FUO - may be recurrence of same diagnosis
Previously diagnosed conditions: malignancy, rheumatic fever, valve disease that predisposes to endocarditis
Verification of fever and pattern of fever
- Verification of fever: Often overlooked step
- In some series, up to 30% referred for FUO where determined to NOT have fever
- Request fever log or have patient take temperatures at home
- Fever patterns - use of patterns to narrow differential:
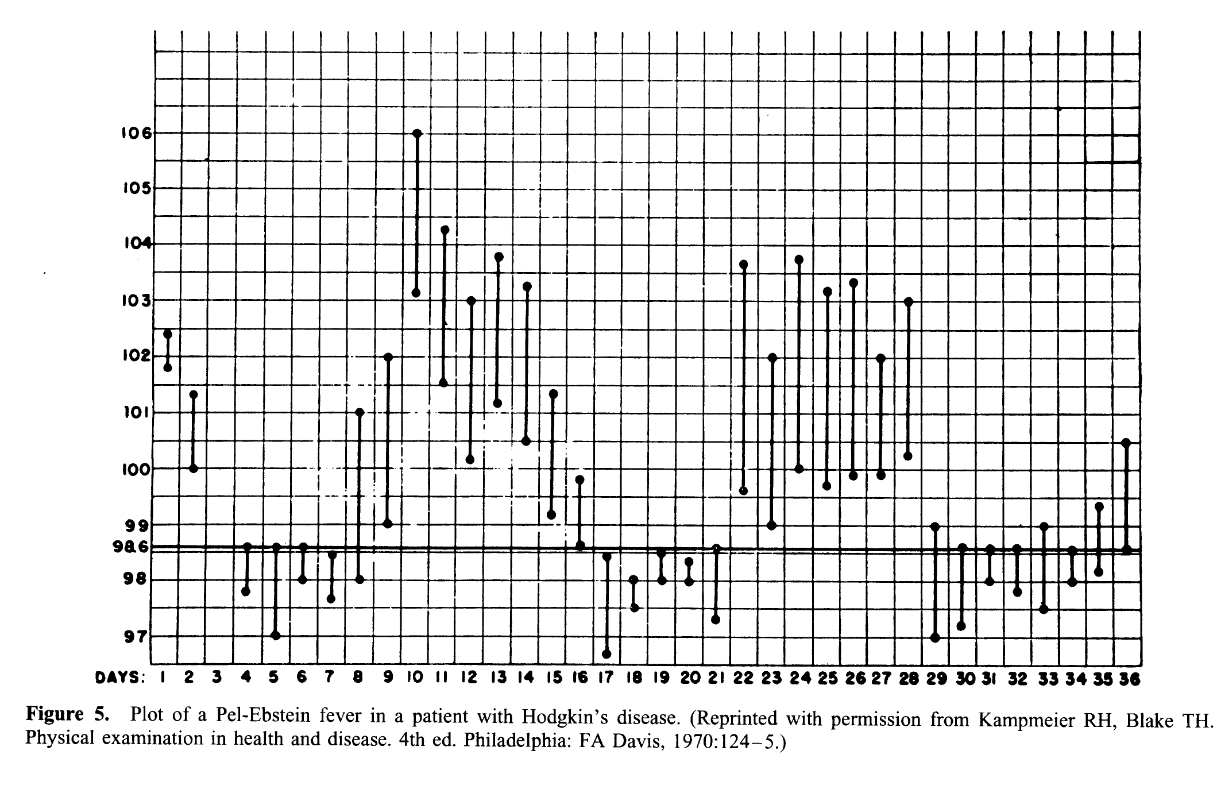
- Continuous, remittent, intermittent, hectic (Charcot’s), quotidian, quartan, biphasic (saddleback), Pel-Ebstein
- Factors affecting fever pattern:
- Hydration status, ambient temperature
- Accuracy of temperature measurements (different sites)
- Antipyretics and corticosteroids suppress fever
- Blood transfusions and other medical interventions
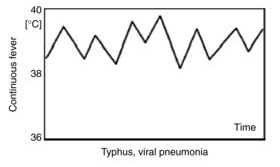
Continuous sustained fever
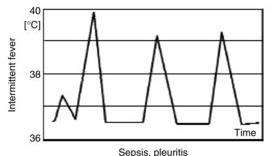
Intermittent (quotidian) fever
- Intermittent fever with wide fluctuations, usually normal or low in the morning and peaking between 4:00 and 8:00 PM
- Localized pyogenic infections and bacterial endocarditis with chills and leukocytosis
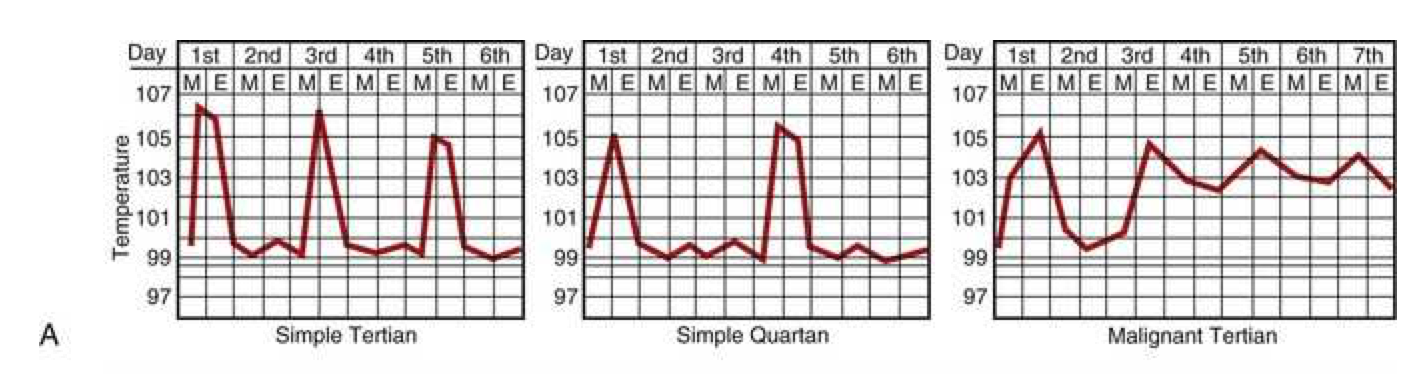
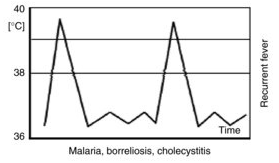
- Malaria may present with daily (quotidian), every 3rd day (tertian), or every 4th day (quartan) patterns
- Double quotidian pattern (two daily spikes) seen with:
- Salmonellosis
- Miliary tuberculosis
- Double malarial infections (>1 species)
- Gonococcal and meningococcal endocarditis
Malaria fever - Paroxysmal patterns
Febrile paroxysms may occur every other day for P. vivax, P. ovale, and P. falciparum (tertian fever) and every third day for P. malariae (quartan fever).
Paroxysms occurring at regular intervals are more common in P. vivax or P. ovale than P. falciparum. With improvements in early diagnosis and treatment, this traditional description of cyclic fever is seen infrequently.
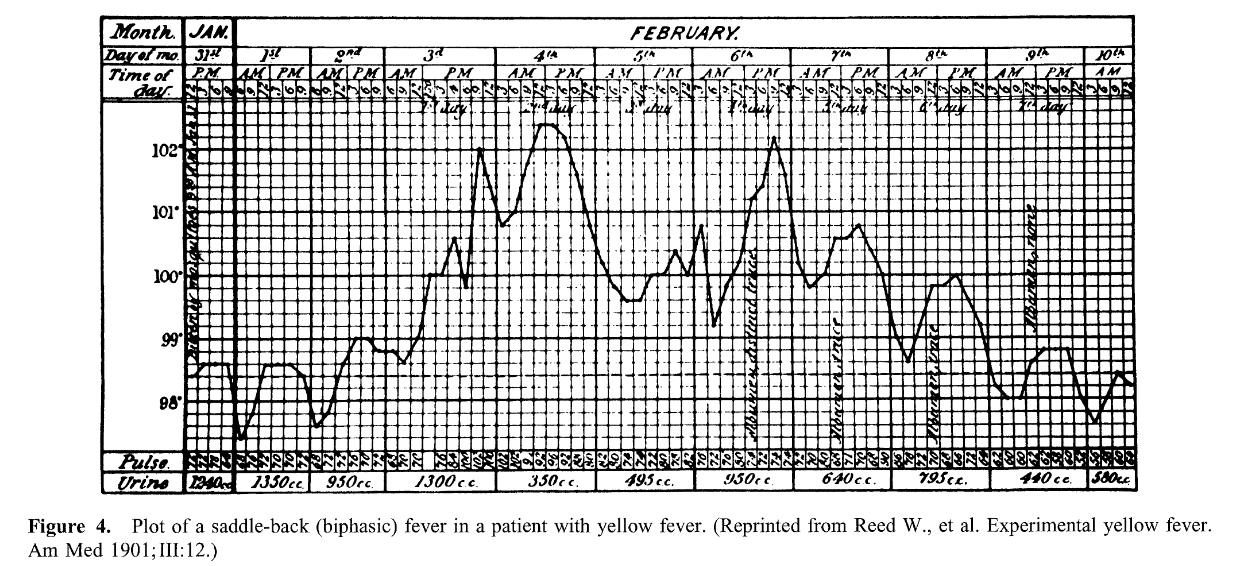
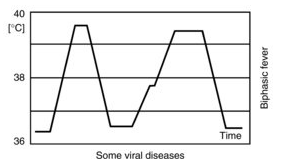
Saddle-back (biphasic) fever
Intermittent hectic (Charcot’s) fever
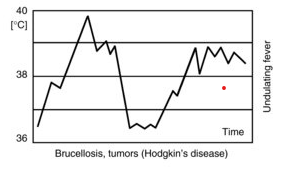
Undulating (Pel-Ebstein) fever
Typus Inversus
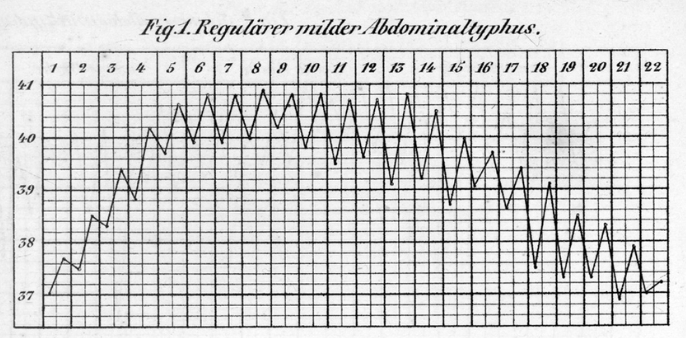
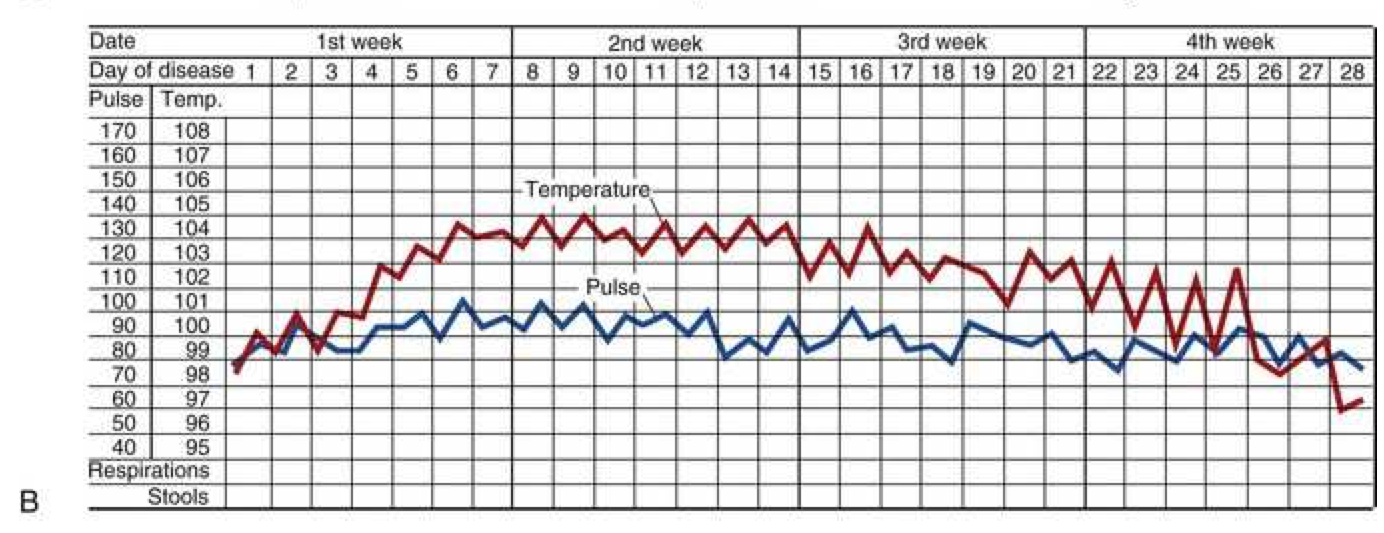
Typhoid fever - Step-ladder fever
Jarisch-Herxheimer reaction
Definition: Sharply increased elevation of temperature with rigors, chills, and constitutional symptoms occurring within hours of starting antibiotic therapy
Mechanism: Lysis of spirochetes (or other organisms) releases endotoxin triggering acute inflammatory response
Organisms associated:
- Primary or secondary syphilis
- Leptospirosis
- Tick-borne relapsing fever (Borrelia)
- Tetracycline or chloramphenicol therapy for acute brucellosis
Clinical significance: Can be severe enough to cause hemodynamic compromise; does not indicate treatment failure
Physical examination
- Some signs are subtle and may require repeated exams to be appreciated
- Vigorous search for lymphadenopathy (consideration for biopsy)
- Careful examination of fundi, oropharynx, temporal arteries, abdomen, spleen, joints, skin, nails, genitalia
| Body site | Physical finding | Diagnosis |
|---|---|---|
| Head | Sinus tenderness | Sinusitis |
| Temporal artery | Nodules, reduced pulsations | Temporal arteritis |
| Oropharynx | Ulceration; tender tooth | Disseminated histoplasmosis, periapical abscess |
| Fundi / conjunctivae | Choroid tubercle, petechiae, Roth’s spot | Disseminated granulomatosis, endocarditis |
| Thyroid | Enlargement, tenderness | Thyroiditis |
| Heart | Murmur | Infective or marantic endocarditis |
| Abdomen | Enlarged iliac lymph nodes, splenomegaly | Lymphoma, endocarditis, disseminated granulomatosis |
| Rectum | Perirectal / prostatic fluctuance, tenderness | Abscess |
| Genitalia | Testicular nodule; epididymal nodule | Periarteritis nodosa; disseminated granulomatosis |
| Lower extremities | Deep venous tenderness | Thrombosis / thrombophlebitis |
| Skin and nails | Petechiae, splinter hemorrhages, subcutaneous nodules, clubbing | Vasculitis, endocarditis |
Laboratory investigations
“The cause of FUO is more frequently a common disease presenting in an atypical fashion than a rare disease presenting in a typical fashion.”
Multiple diagnostic algorithms exist in literature
Must be selectively applied or will result in excessive unfocused diagnostic testing
- False positives lead to misdiagnosis
- Misguided treatment plans
“Sutton’s Law” - pursue most likely diagnosis first based on history and epidemiology
History and physical exam (most important) should guide choice and sequence of tests
Examples of potential diagnostic clues (1/3)
| Etiology | Historical clues | Physical clues |
|---|---|---|
| Anaplasmosis | Ixodes tick bite; outdoor activity in North Central / Eastern US | Fever, headache, arthralgia, myalgia, pneumonitis, thrombocytopenia, lymphopenia, ↑ liver enzymes |
| Babesiosis | Ixodes tick bite; outdoor activity in Northeastern US | Arthralgias, myalgias, relative bradycardia, hepatosplenomegaly, anemia, thrombocytopenia, ↑ liver enzymes |
| Bartonellosis | Travel to Andes (Oroya fever; B. bacilliformis); homelessness (B. quintana); scratch from infected kitten/cat (B. henselae) | Conjunctivitis, retro-orbital pain, anterior tibial bone pain, macular rash, nodular plaque lesions, regional lymphadenopathy |
| Blastomycosis | Contact with soil near Mississippi/Ohio River valleys, Saint Lawrence River, or Great Lakes; exposure to infected dogs | Arthritis, atypical pneumonia, pulmonary nodules, ARDS, verrucous/nodular/ulcerative skin lesions, prostatitis |
| Brucellosis | Contact with/consumption of products from infected goats, pigs, camels, yaks, buffalo, cows; abattoir work | Arthralgias, hepatosplenomegaly, suppurative musculoskeletal lesions, sacroiliitis, spondylitis, uveitis, hepatitis, pancytopenia |
| Coccidioidomycosis | Exposure to soil or dust in the southwestern US | Arthralgias, pneumonia, pulmonary cavities, pulmonary nodules, erythema multiforme, erythema nodosum |
| Ehrlichiosis | Amblyomma, Dermacentor, or Ixodes tick bite; outdoor activity in midwestern / southeastern US | Pneumonitis, hepatitis, thrombocytopenia, lymphopenia |
Examples of potential diagnostic clues (2/3)
| Etiology | Historical clues | Physical clues |
|---|---|---|
| Enteric fever (Salmonella Typhi) | Recent travel to endemic country; consumption of potentially contaminated food or water | Headache, arthritis, abdominal pain, relative bradycardia, hepatosplenomegaly, leukopenia |
| Histoplasmosis | Exposure to bat/blackbird excreta in roosts, chicken houses, or caves; Ohio and Mississippi River valleys | Headache, pneumonia, pulmonary cavities, mucosal ulcers, adenopathy, erythema nodosum, erythema multiforme, hepatitis, anemia, leukopenia, thrombocytopenia |
| Leptospirosis | Occupational exposure in sewers, rice/sugar cane fields, abattoirs; recreational water sports; contact with contaminated water or infected dogs | Bitemporal/frontal headache, calf and lumbar muscle tenderness, conjunctival suffusion, hepatic and renal failure, hemorrhagic pneumonitis |
| Leishmaniasis (visceral) | Recent travel to sand fly–endemic areas | Hepatosplenomegaly, lymphadenopathy, hyperpigmentation of face/hands/feet/abdomen (kala azar) |
| Malaria | Recent travel to endemic areas in Asia, Africa, or Central/South America | Fever, headaches, nausea, vomiting, diarrhea, hepatosplenomegaly, anemia |
| Psittacosis (Chlamydia psittaci) | Contact with birds, especially psittacine birds | Fever, pharyngitis, hepatosplenomegaly, pneumonia, blanching maculopapular eruptions, erythema multiforme, erythema marginatum, erythema nodosum |
Examples of potential diagnostic clues (3/3)
| Etiology | Historical clues | Physical clues |
|---|---|---|
| Q fever (Coxiella burnetii) | Farm, veterinary, or abattoir work; unpasteurized milk; contact with infected sheep, goats, or cattle | Atypical pneumonia, hepatitis, hepatomegaly, relative bradycardia, splenomegaly |
| Rat-bite fever (Streptobacillus moniliformis) | Recent bite/scratch from rat, mouse, or squirrel; ingestion of food/water contaminated with rat excrement | Headaches, myalgias, polyarthritis, maculopapular/morbilliform/petechial/vesicular/pustular rash over palms, soles, and extremities |
| Relapsing fever (Borrelia recurrentis) | Poverty/crowding/poor sanitation (louse-borne); camping in the Grand Canyon (tick-borne) | High fever with rigors, headache, delirium, arthralgias, myalgias, hepatosplenomegaly |
| Rocky Mountain spotted fever | Outdoor activity in South Atlantic or southeastern US; Dermacentor tick bites | Headache, petechial rash on extremities, palms, and soles |
| Tuberculosis | Contact with TB; immigration from endemic country; homeless shelter or healthcare facility exposure | Night sweats, weight loss, atypical pneumonia, cavitary pulmonary lesions |
| Tularemia | Bites from Amblyomma or Dermacentor ticks, deer flies, or mosquitoes; direct contact with rabbits, squirrels, deer, raccoons, cattle, sheep, or swine | Ulcerated skin lesion at bite site, pneumonia, relative bradycardia, lymphadenopathy, conjunctivitis |
| Whipple’s disease (Tropheryma whipplei) | Potential association with exposure to sewage | Chronic diarrhea, arthralgia, weight loss, malabsorption, malnutrition |
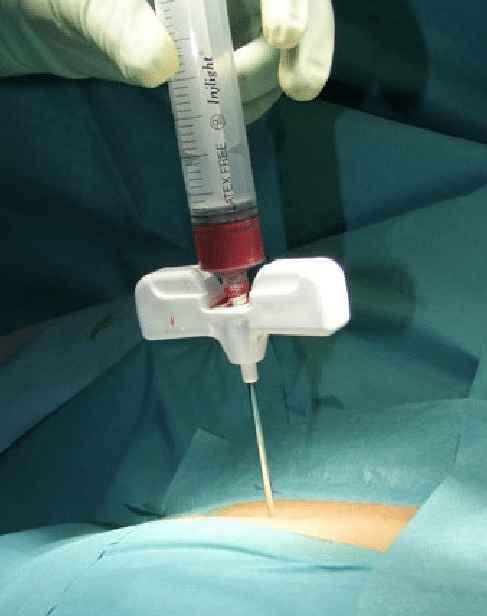
Bone marrow biopsy
- Diagnostic yield: ~25% in two case series; especially valuable for:
- Granulomatous infections (TB, histoplasmosis, sarcoidosis)
- Hematologic malignancies (leukemia, lymphoma)
- Patients with abnormal CBC (anemia, thrombocytopenia, leukopenia)
- Organisms identified:
- Mycobacterium tuberculosis
- Fungi (Histoplasma, Cryptococcus, Coccidioides)
- Intracellular bacteria (Brucella, Bartonella)
- Consider: When fever pattern and presentation suggest granulomatous disease or hematologic malignancy
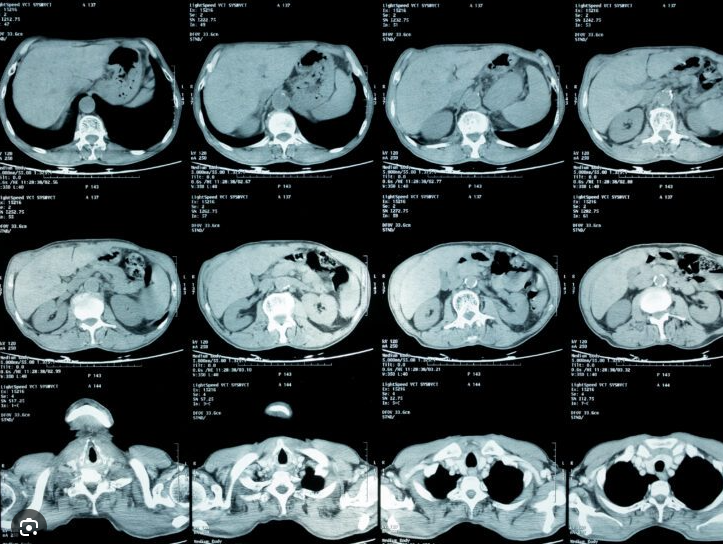
Imaging studies
Generally low diagnostic yield without localizing symptoms
CT of abdomen and chest
Ultrasound of gallbladder and hepatobiliary systems
CT pulmonary angiogram (for pulmonary embolis)
MRI for CNS, spleen, lymph nodes, aorta (vasculitis)
Indium 111-tagged white blood cell scan (becoming less common)
Gallium-67 scan (largely replaced by PET-CT)
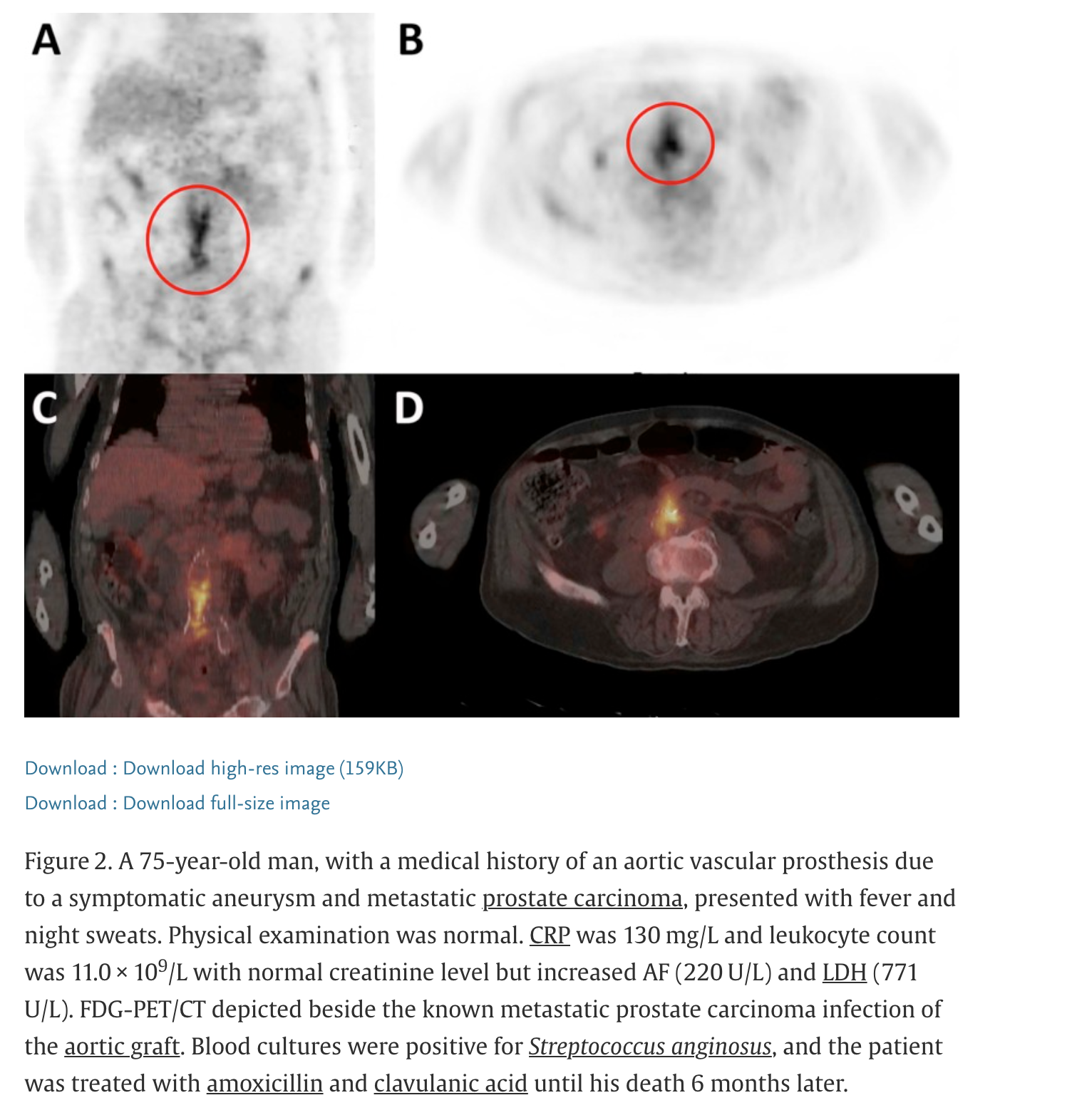
PET-CT: Superior sensitivity for inflammatory and malignant processes
¹⁸F-fluorodeoxyglucose (FDG) positron emission tomography
Invasive diagnostic procedures
- Biopsy techniques:
- Excisional biopsy (lymph node)
- Needle biopsy (liver, kidney, muscle)
- Laparotomy or laparoscopy for direct visualization
- Diagnostic yield:
- Biopsy yields diagnosis in < 50% of cases
- Average 2-3 biopsies needed to establish diagnosis
- Indications:
- Abnormal imaging or physical exam findings
- Lymphadenopathy suitable for lymph node biopsy
- Suspected granulomatous disease
- Persistent fever despite extensive workup
Treatment Section
A fundamental principle in classic FUO:
Therapy should be withheld until the cause of fever is determined
Diagnostic summary and approach
When is immediate treatment indicated?
- Suspected temporal arteritis
- Empirical corticosteroids to prevent vascular complications (blindness, stroke)
- Do not wait for biopsy confirmation if clinical suspicion high
- Febrile neutropenia or severe immunocompromise
- High prevalence of serious bacterial infections
- Broad-spectrum antimicrobial therapy with anti-pseudomonas coverage after appropriate cultures obtained
- No delay for culture results
- Select cases with strong clinical suspicion:
- Anti-mycobacterial therapy in suspected TB (especially if respiratory symptoms)
- Targeted therapy based on epidemiology and presentation
Prognosis
Determined by: The underlying cause of fever and nature of underlying disease(s)
Poor prognosis: Elderly patients with malignant neoplasms
Diagnostic delay worsens prognosis in:
- Intra-abdominal infections (perforation, sepsis)
- Miliary tuberculosis
- Disseminated fungal infections
- Recurrent pulmonary emboli
Favorable outlook: Patients with undiagnosed FUO after extensive evaluation
- Most experience resolution of fever within 4 weeks without sequelae
- 5-year mortality rate: 3.2% for undiagnosed FUO
- Superior to patients with diagnosed malignancy as the underlying cause