%%{init: {
'theme': 'base',
'themeVariables': {
'fontFamily': "Arial, sans-serif",
'fontSize': '24px',
'primaryColor': '#cfe2f3',
'primaryTextColor': '#000000',
'primaryBorderColor': '##ffffff',
'lineColor': '#000000',
'secondaryColor': '#d9ead3',
'tertiaryColor': '#fce5cd'
}
}
}%%
timeline
section Suspension era- <br>TDM essential for all patients
2006: Suspension formulation approved by the FDA, EMA
2007: Prophylaxis and refractory IA data: Cornely et al. NEJM: Ullmann et al. NEJM: Walsh et al. CID : Treatment > 1000 ng/mL
2010: TDM targets- prophylaxis > 700 ng/mL: Jang et al. CP&T
2012: "Real-life" validation of 500-700 ng/mL: Dolton et al. AAC: Many other single and multi centre retrospective studies : TDM incorporated in treatment guidelines
section Gastro-resistant tablet/IV era- <br> TDM still essential?
2013: FDA/EMA approval of posaconazole ER tablets
2015: FDA/EMA approval of posaconazole IV formulation: Cornely et al. JAC
2020: Toxicity threshold? : Nguyen et 2020: Pseudo- <br>hypo- <br>aldosteronism, hepatotoxicity > 3000 ng/mL
2021: Phase III treatment data: Maertens et al. Lancet
2023: No observed ER relationship: Maertens et al. <br>Clin Drug Investig
section Targeted TDM- <br> which patients?
2025: Wide PK variability in ICU : Roberts et al. <br> SAFE-ICU <br> Inten Care Med
Posaconazole therapeutic drug monitoring:
Precision vs. pragmatism?
Russell E. Lewis, Pharm.D., FCCP
Associate Professor of Infectious Diseases
Department of Molecular Medicine
University of Padua
Disclosures
Royalities: UptoDate
Research support: Gilead Inc.
Speaking: Avir, Gilead, F2G, Pfizer
Advisory: Gilead, F2G, Synexis, Basilea, Cidara
Timeline of posaconazole TDM
Case #1: Posaconazole prophylaxis post HSCT
56 year-old female patient with AML undergoing haploidentical HSCT with myeloablative BuFLU -thiotepa
Prophylaxis:
Micafungin 50 mg/day starting at neutropenia onset until completion of PTCy on days +3,+4
Posaconazole ER tablets 300 mg BID day #1, then 300 mg daily starting day+5
Patient is also started on letermovir, tacrolimus, mycophenolate, valacyclovir
On day +9 of transplant when patient has ANC 50 and mucositis (still can swallow solid food), a posaconazole trough concentration is ordered
Result= 450 ng/mL (Target > 700 ng/mL)
What should we do?
- No change in dose, repeat TDM in 7 days
- Increase dose of posaconazole ER tablets to 400 mg daily, repeat TDM in 7 days
- Switch to IV posaconazole (if available)
- Switch to oral isavuconazole or IV isavuconazole
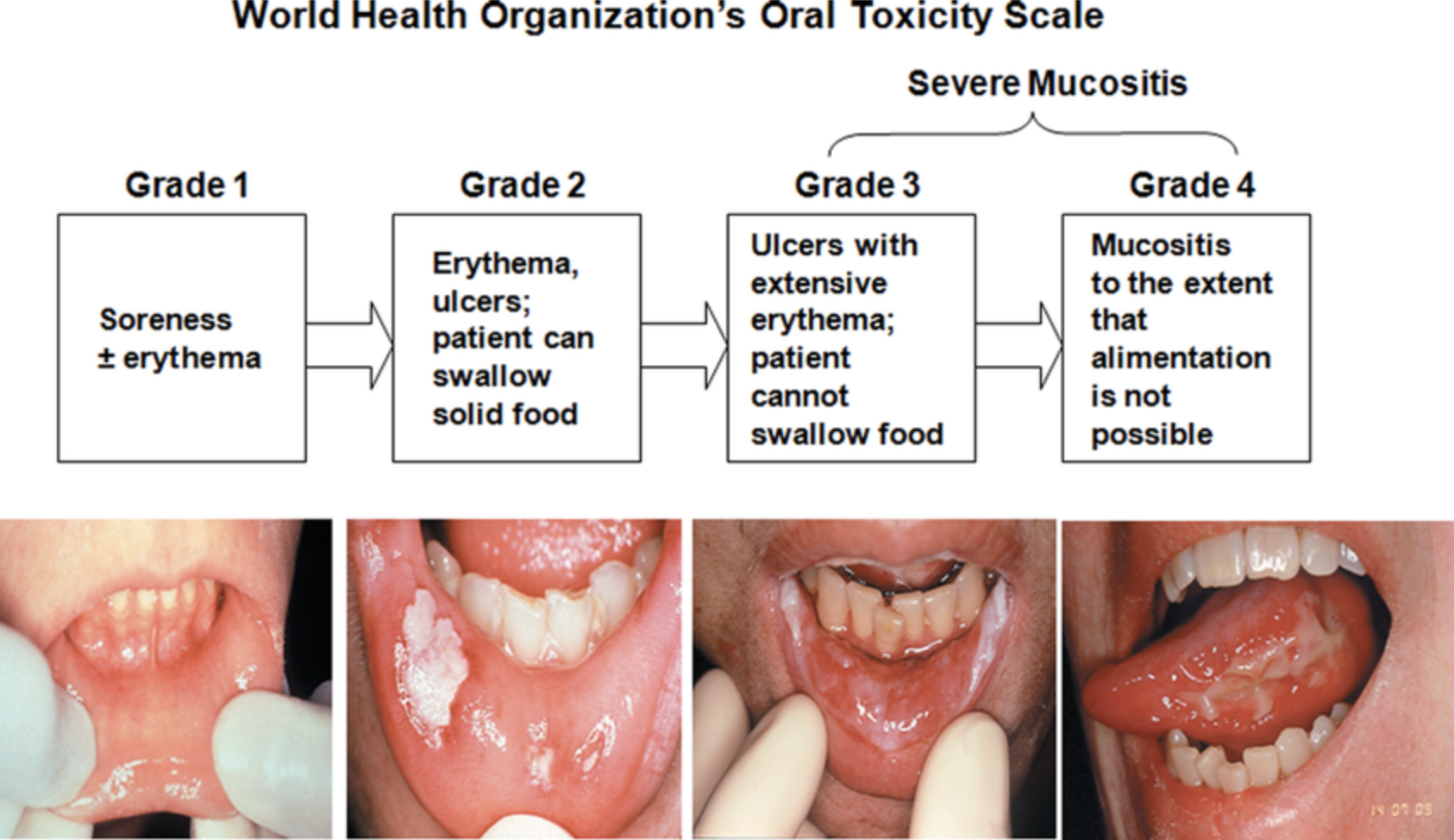
Mucositis grading system
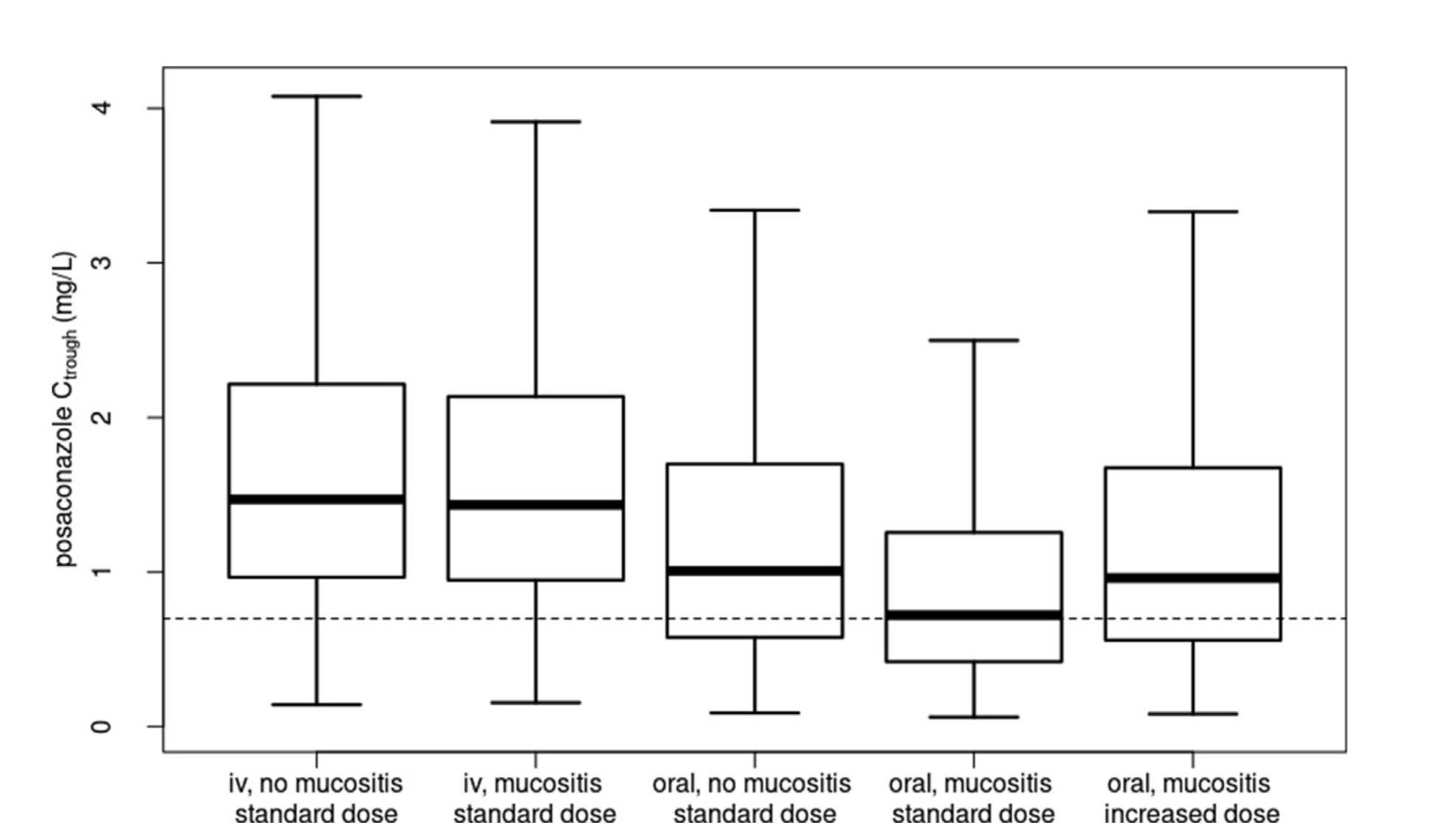
Reduced bioavailibility in the setting of
intestinal mucositis
Dose-escalation during mucositis?
Use IV formulation (if available)
Pragmatic increase of loading dose to 400 mg BID on day 1, then 400 mg daily
Higher doses are generally well-tolerated during treatment of triazole-resistant isolates
- Hepatotoxicity, pseudohyperaldosteronism (serum levels > 3000 ng/mL)
Not always easy to predict who develops severe mucositis-
citrulline biomarker not routinely used in clinic- Follow oral food intake/ Visual assessment?
Will higher doses reduce the risk of breakthrough fungal disease?
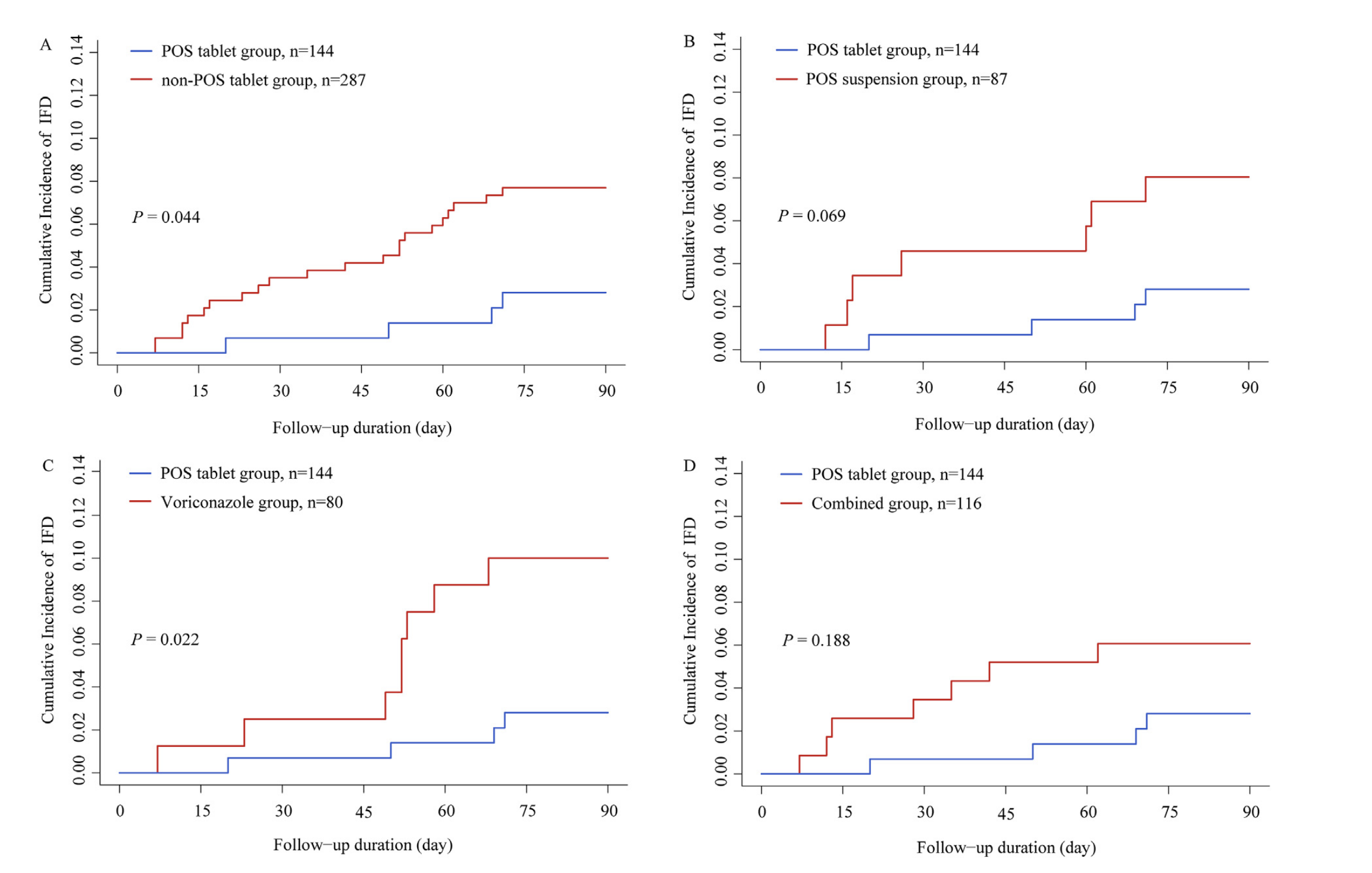
Delayed attainment of target posaconazole
trough concentrations after HSCT
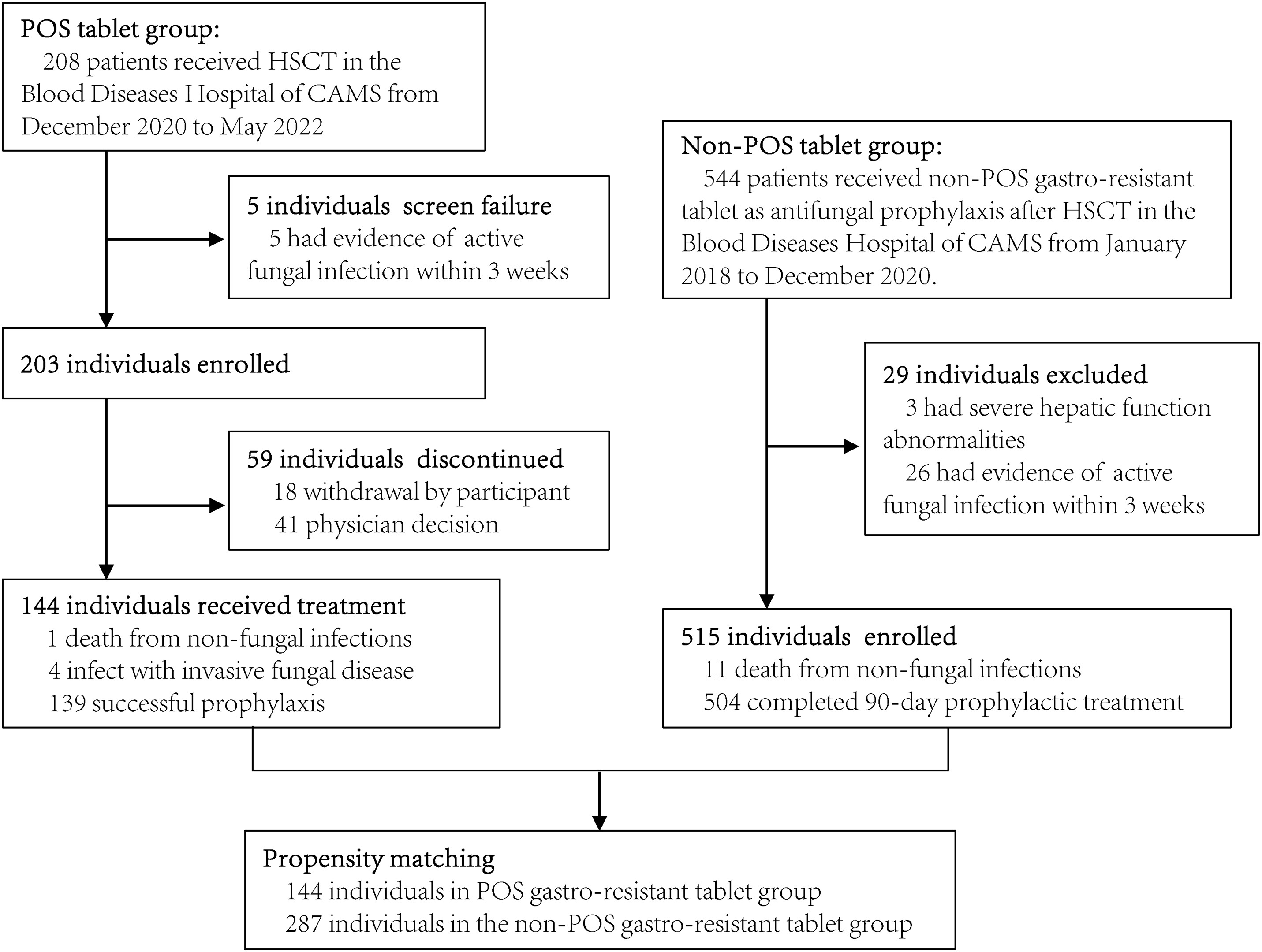
Study design
Primary study endpoint: Incidence of breakthrough IFIs
Delayed attainment of target posaconazole
trough concentrations after HSCT
Delayed attainment of target posaconazole
trough concentrations after HSCT
Case #2: Posaconazole precision dosing?
- 65 year-old patient with AML, fever, severe respiratory difficulty following consolidation chemotherapy -absolute neutrophil count 250
- Patient was receiving voriconazole, but switched to isavuconazole following development of mental status changes
- Influenzae A positive- oseltamivir
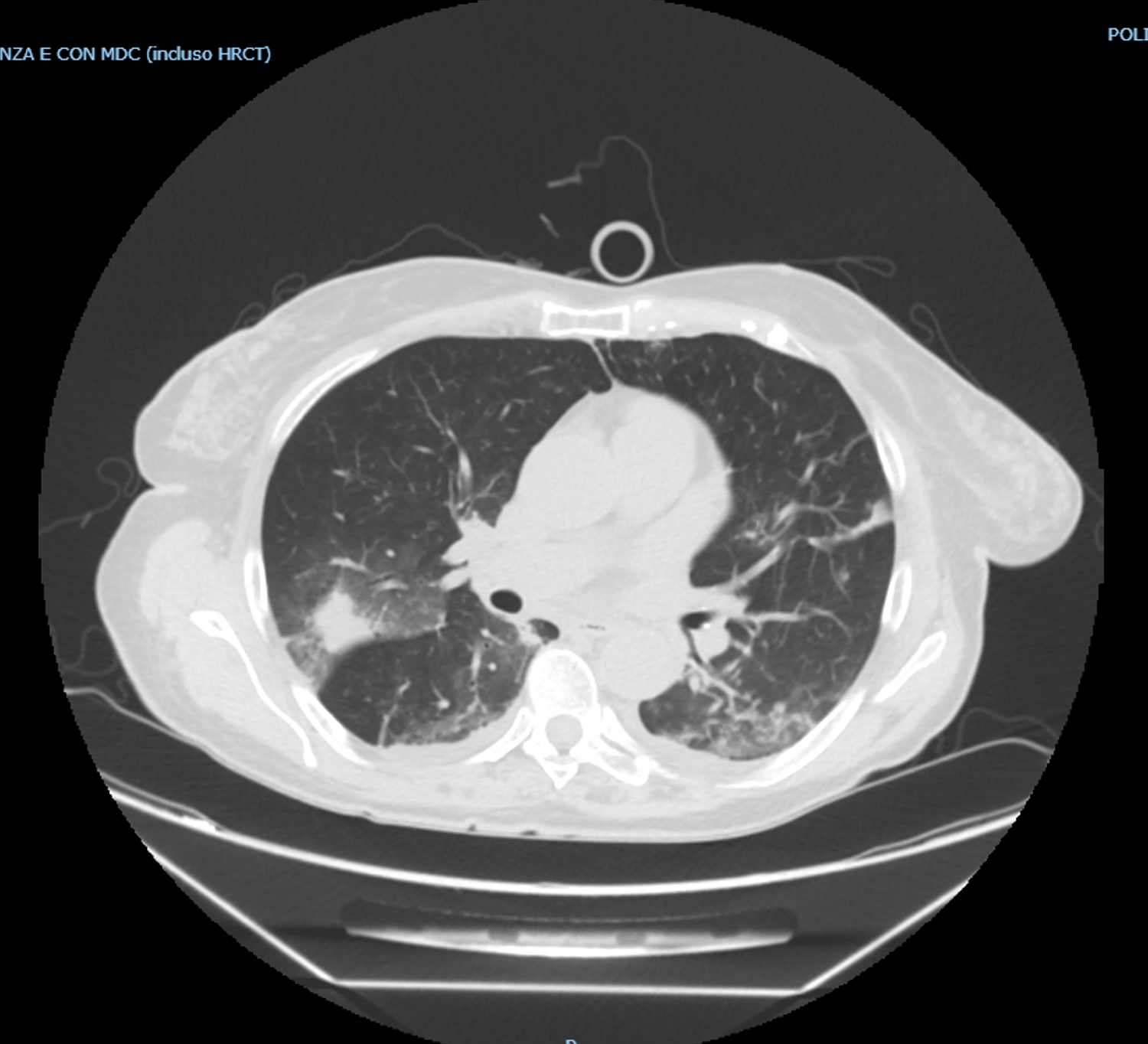
- Bronchoscopy positive for Aspergillus terreus, galactomannan 3.1
Aspergillus terreus antibiogram
| Antifungal | MIC (mg/L) | EUCAST interpretation |
|---|---|---|
| Amphotericin B | 4 | IE |
| Anidulafungin | >4 | IE |
| Caspofungin | >4 | IE |
| Micafungin | >4 | IE |
| Isavuconazole | 2 | R |
| Voriconazole | 1 | IE, ECOFF=2 |
| Posaconazole | 0.06 | S, breakpoint= 0.25 |
Clinical management
- Patient is switched to posaconazole ER tablets 300 mg BID day 1, then 300 mg daily
- After 5 days a posaconazole trough is sent, result =710 ng/mL
- Patient is clinically stable, improving respiratory status and afebrile
What do you do?
- No change in dose, repeat TDM in 7 days
- Increase dose of posaconazole ER tablets to 400 mg daily, repeat TDM in 7 days
- Use combination therapy
- Explore options for olorofim, fosmanogepix?
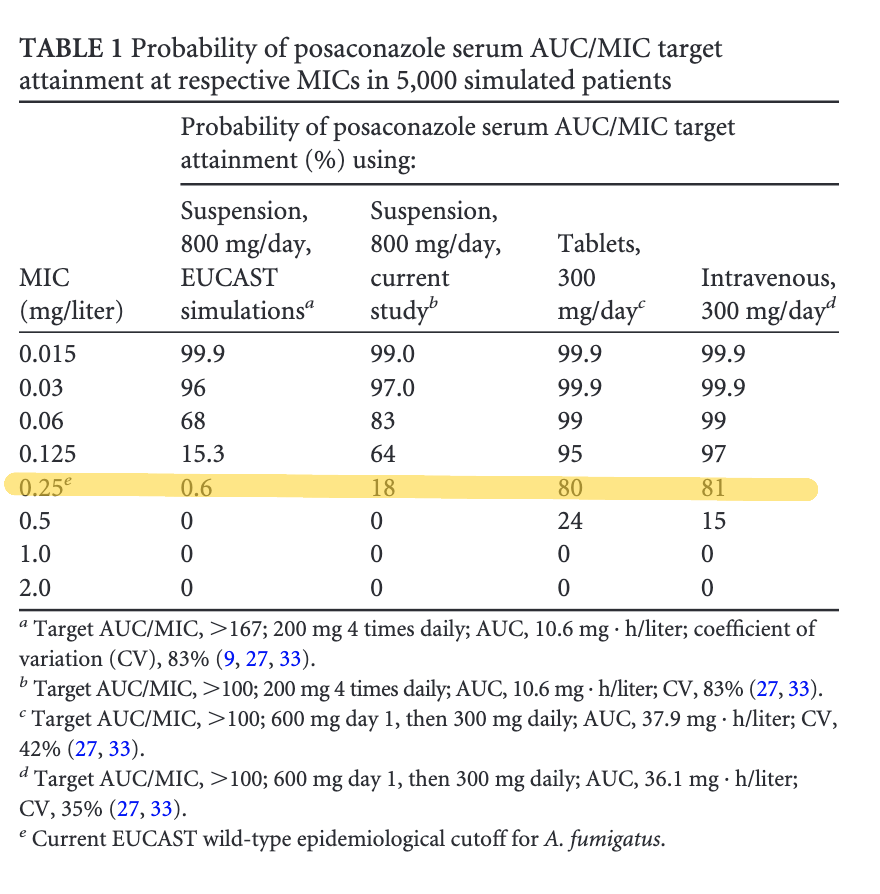
Do we need to increase the dose?
Or just assume the MIC is 0.25 mg/L?
One other twist….
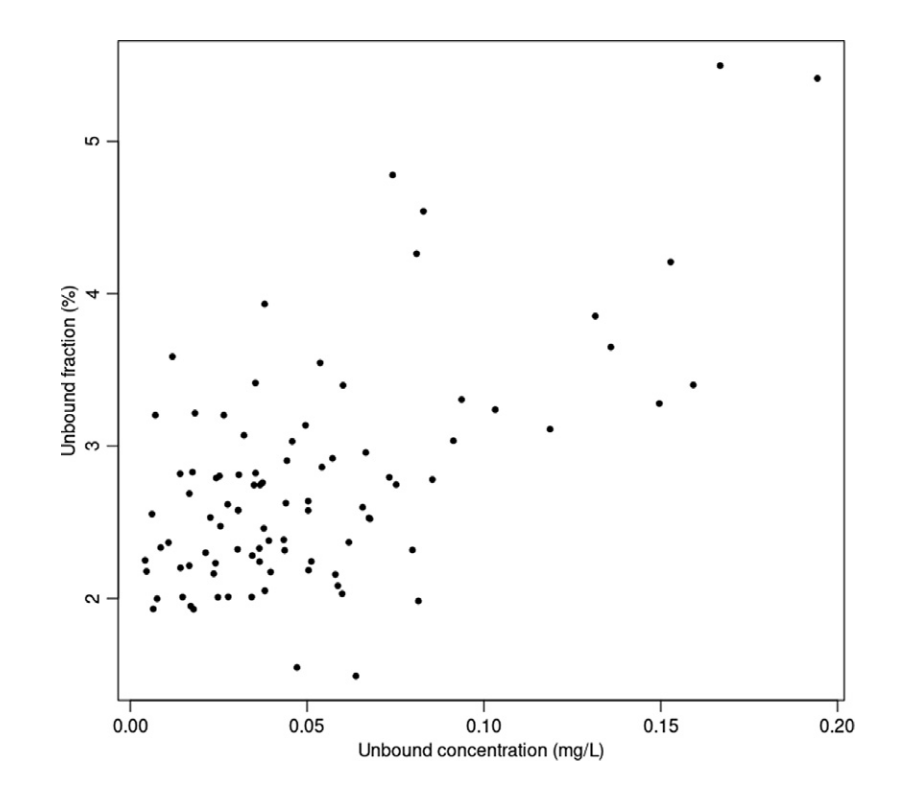
- Patient has a serum albumin of 2.2 mg/dL
- Trough treatment target 1000-3750 ng/L: free drug (99% protein bound) 10-37.5 ng/mL
- In theory, her total serum posaconazole concentrations are decreased, but unbound concentration is the same and percent unbound actually increases ~ 2 fold
- Because TDM results are not reported as free drug, should we “correct” posaconazole results for low albumin?
Corrected total concentration of posaconazole
in this patient?
\[ C_{corr}= C_{obs}\frac{1}{\left( 0.01 + 0.99\frac{Alb}{4.4} \right)} \]
\[ C_{corr}= C_{obs}\frac{4.4}{Alb} \]
\[ C_{corr}= 1400 ng/mL \]